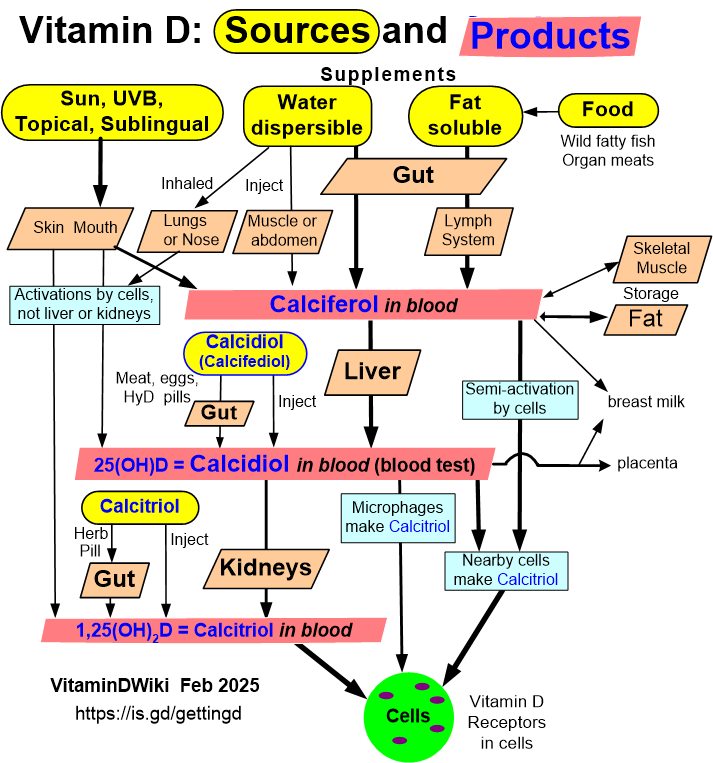
35 tissues activate Vitamin D locally (Kidney not required)
3 Types of Vitamin D
7+ Vitamin D Genes
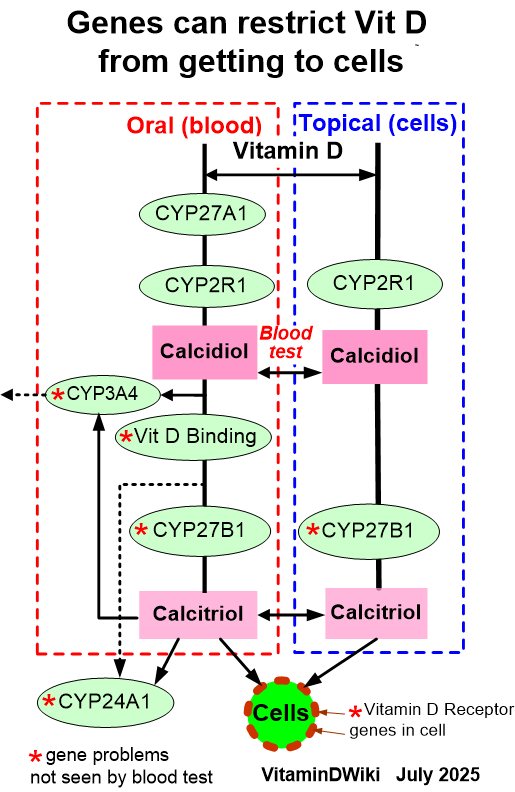
Related in VitaminDWiki
Extrarenal CYP27B1: Local Calcitriol Activation in Humans
Claude AI - Deep Research - May 2026
Summary Table — Conditions Mapped to Calcidiol-Substrate Mechanism
| Condition | Proposed calcidiol-substrate mechanism | Strength of evidence | Approximate 25(OH)D threshold below which risk rises |
|---|---|---|---|
| Preeclampsia | Low maternal 25(OH)D → reduced placental/decidual 1,25(OH)₂D → impaired Treg induction, spiral-artery remodeling, anti-angiogenic dysregulation | Strong observational (Bodnar, Achkar, Baca); RCT signal mixed (MAVIDOS post-hoc null for BP; VDAART secondary positive) | <50 nmol/L (20 ng/mL); plateau benefit ≥75 nmol/L (30 ng/mL) |
| Gestational diabetes | Substrate limitation of pancreatic β-cell and adipose VDR/CYP27B1 signaling; impaired placental insulin-sensitizing milieu | Moderate (consistent meta-analyses, OR 1.15–1.61) | <50 nmol/L |
| Preterm birth | Reduced placental cathelicidin/HBD2 (ascending infection); altered local immune tolerance; cervical-stromal effects | Strong (multiple meta-analyses + Hollis–Wagner RCTs + McDonnell post-hoc) | ≥40 ng/mL (100 nmol/L) for max protection |
| SGA / IUGR | Impaired trophoblast invasion and spiral-artery remodeling via local 1,25(OH)₂D and G-CSF | Moderate | <50 nmol/L |
| Recurrent miscarriage | Decidual VDR/CYP27B1 dysfunction; impaired immune tolerance; impaired decidualization | Moderate–strong (Tamblyn 2022 OR 1.94; Chen 2022 OR 4.02) | <50 nmol/L; possibly <75 nmol/L |
| Bacterial vaginosis | Reduced vaginal/cervical epithelial cathelicidin/HBD2 from local CYP27B1 | Strong dose–response | Plateau benefit at 25(OH)D ~80 nmol/L (32 ng/mL); steep risk <30 ng/mL |
| Intrauterine infection / chorioamnionitis | As BV mechanism plus trophoblast cathelicidin | Moderate | <30 ng/mL |
| Offspring asthma/wheeze | Maternal 25(OH)D substrate for fetal lung-resident immune cell calcitriol; sphingolipid/ORMDL3 axis | Moderate (VDAART positive at 4400 IU; non-significant at primary 3-y endpoint) | Maternal ≥30 ng/mL desirable |
| Offspring bone mass | Maternal substrate for placental and fetal CYP27B1; calcium handoff | Strong (MAVIDOS winter-birth signal; 4-y BMD) | Maternal ≥50 nmol/L; winter-birth particular risk |
| Offspring neurodevelopment | Brain CYP27B1 active mid-gestation; calcitriol regulates neurogenesis | Mixed (positive ALSPAC autism; null 2024 FMC schizophrenia) | <25–30 nmol/L (most consistent) |
| Offspring MS | Substrate for neural/immune CYP27B1 during developmental window | Moderate (FMC 2017) | <30 nmol/L |
| Tuberculosis | Macrophage TLR2/1-induced CYP27B1 → cathelicidin (LL-37) → mycobacterial killing | Mechanistic strong (Liu/Modlin 2006); RCT evidence for prevention null/weak; adjunctive modest | Cathelicidin induction inadequate <20 ng/mL |
| Acute respiratory infections | Macrophage and epithelial CYP27B1 → cathelicidin + IL-37; restrained inflammation | Strong (Martineau IPD meta-analyses; effect concentrated in <25 nmol/L; daily/weekly dosing only) | <25 nmol/L for largest benefit |
| COVID-19 | Substrate for pulmonary/macrophage CYP27B1; calcifediol bypasses delay | Suggestive (Castillo RCT large effect; subsequent cholecalciferol RCTs mixed) | <20 ng/mL high-risk; calcifediol restores rapidly |
| Sepsis / ICU | Substrate for immune-cell intracrine signaling; reduced cathelicidin and tissue repair | Observational strong; supplementation RCT mixed (VITdAL-ICU positive in deficient subgroup) | <12 ng/mL severe; <20 ng/mL elevated |
| MS | Substrate for myeloid/CNS CYP27B1; central tolerance | Strong observational (Munger 2006, 2017; free 25(OH)D Grut 2022) | <50 nmol/L (rising risk); optimum likely ≥75 nmol/L |
| Type 1 diabetes | β-cell and immune CYP27B1; modulates Tregs and IFN-α response | Moderate (cohort) | <50 nmol/L |
| RA, IBD, lupus | Synovial/intestinal/dendritic-cell local activation | Moderate (cohorts + VITAL autoimmune signal HR 0.78) | <30 ng/mL |
| Colorectal, breast, prostate cancer mortality | Local tumor and stromal CYP27B1 → anti-proliferative, pro-differentiation, pro-apoptotic | Moderate (VITAL: cancer mortality ↓17%, advanced cancer ↓38%) | ≥30–40 ng/mL likely optimal for mortality benefit |
| Cardiovascular / endothelial dysfunction | Endothelial and VSMC CYP27B1 | Weak–moderate (Mendelian randomization mostly null) | <20 ng/mL |
| Cognitive decline / dementia | Hippocampal CYP27B1, neurosteroid effects | Moderate observational | <50 nmol/L |
| Periodontal disease | Gingival CYP27B1; cathelicidin | Moderate | <30 ng/mL |
| Atopic dermatitis / psoriasis | Keratinocyte CYP27B1; topical analogs bypass deficiency | Mechanistic strong; topical calcipotriol effective | Systemic <30 ng/mL contributes |
Master Summary Table
Evidence grade key:
- mRNA = transcript detected;
- P = protein (IHC/Western);
- A = enzymatic conversion of 25(OH)D₃ → 1,25(OH)₂D₃ demonstrated in vitro;
- S = 1,25(OH)₂D detectable in cell-culture supernatant or causing systemic hypercalcemia;
- KO = knockout phenotype supports physiological role.
| System | Tissue / cell | Evidence | Pivotal citation(s) | Proposed local role |
|---|---|---|---|---|
| Reproductive | Placenta — syncytio- & cytotrophoblast | mRNA+P+A+S+KO | Weisman 1979 Nature; Zehnder 2002 Am J Pathol; Diaz 2000 J Clin Endocrinol Metab 85:2543 | Pregnancy Ca²⁺ economy; trophoblast invasion; immune tolerance |
| Reproductive | Decidua | mRNA+P+A | Zehnder 2002 Am J Pathol 161:105 | Maternal–fetal interface tolerance |
| Reproductive | Ovary — granulosa, theca, luteal | mRNA+P+A | Fischer 2009 Mol Cell Endocrinol; Brożyna 2014 Oncol Rep 33:599 | Follicular growth, steroidogenesis |
| Reproductive | Testis — Leydig, Sertoli, spermatogenic | mRNA+P | Blomberg Jensen 2010 Hum Reprod 25:1303 | Spermatogenesis, calcium signaling |
| Reproductive | Endometrium | mRNA+P+A | Vienonen 2004; Agic 2007; Becker 2007 Mol Cell Endocrinol | Cycle-dependent proliferation control |
| Reproductive | Cervix | mRNA+P | Friedrich 2002 Anticancer Res 22:1849 | Differentiation control |
| Reproductive | Prostate epithelium | mRNA+P+A+S | Schwartz 1998 Cancer Epidemiol Biomarkers Prev; Hsu 2001 Cancer Res 61:2852; Chen 2003 J Cell Biochem 88:315 | Tumor-suppressor / antiproliferative |
| Reproductive | Breast (mammary epithelium) | mRNA+P+A+S | Kemmis 2006 J Nutr 136:887; Segersten 2005 Breast Cancer Res 7:R980 | Differentiation, lactation, tumor suppression |
| Immune | Pulmonary alveolar macrophages (sarcoid/TB) | mRNA+P+A+S | Adams 1983 J Clin Invest 72:1856; Liu 2006 Science 311:1770 | Cathelicidin/β-defensin antimicrobial; hypercalcemia in disease |
| Immune | Monocyte-derived macrophages | mRNA+P+A | Kreutz 1993; Stoffels 2006 Endocrinology | TLR-induced antimicrobial response |
| Immune | Dendritic cells (myeloid) | mRNA+P+A | Hewison 2003 J Immunol 170:5382; Sigmundsdottir 2007 Nat Immunol 8:285; Kundu 2014 Eur J Immunol | Tolerogenic DC phenotype; T-cell homing |
| Immune | Activated CD4⁺/CD8⁺ T cells | mRNA+P+A | Sigmundsdottir 2007; Ooi 2014 Mol Cell Endocrinol 388:74 | Autocrine Treg differentiation; Th1/Th17 suppression |
| Immune | Activated B cells | mRNA+P | Chen 2007 J Immunol 179:1634; Heine 2008 Eur J Immunol | IgE suppression, plasma cell regulation |
| Immune | Lymph node (CD68⁺ macrophages, granulomata) | P+A | Zehnder 2001 JCEM 86:888 | Antimicrobial; pathology in sarcoid |
| Immune | Thymus, spleen, bone marrow stroma | mRNA+P | Zehnder 2001; Bland 2004 J Steroid Biochem Mol Biol | T-cell development; hematopoiesis |
| Skin | Keratinocytes (basal) | mRNA+P+A+S+KO | Bikle 1986 J Clin Invest 78:557; Zehnder 2001; Lehmann 2009 Anticancer Res | Differentiation, barrier, antimicrobial; complete pathway from 7-DHC |
| Skin | Hair follicle (outer root sheath) | P | Zehnder 2001 JCEM 86:888 | Hair-cycling (largely VDR-ligand-independent in KO data) |
| Skin | Melanocytes | mRNA+A | Frankel 1983; Brożyna 2012 Hum Pathol 44:374 | Pigmentation, antiapoptotic; lost in melanoma progression |
| Skin | Sebaceous glands | mRNA+P | Reichrath 2007 Anticancer Res | Sebocyte differentiation |
| GI | Colon enterocytes/colonocytes | mRNA+P+A | Bareis 2002 Exp Cell Res 276:320; Tangpricha 2001 Lancet 357:1673; Zehnder 2001; Liu 2013 J Biol Chem (mouse model) | Antiproliferative; barrier; antimicrobial; tumor suppression |
| GI | Small intestine epithelium | mRNA+P | Theodoropoulos 2001 J Endocrinol Invest | Local Ca²⁺ absorption augmentation |
| GI | Esophagus | mRNA+P | Brożyna 2014; Townsend 2005 J Steroid Biochem Mol Biol | Differentiation; cancer protection |
| GI | Stomach | mRNA | Bareis (review) | Speculative |
| GI | Liver — hepatocytes, Kupffer, hepatic stellate | mRNA+P+A | Gascon-Barré 2003 Hepatology 37:1034 | Antifibrotic on stellate cells |
| GI | Gallbladder epithelium | mRNA+P (limited) | Theodoropoulos 2003 | Cholangiocyte differentiation |
| GI | Pancreas — β-cells, α-cells, ductal | mRNA+P+A | Bland 2004 J Steroid Biochem Mol Biol 89:121; Zehnder 2001 | Insulin secretion modulation; antiapoptotic |
| Cardiovascular | Vascular endothelium (HUVEC, renal artery) | mRNA+P+A+S | Zehnder 2002 J Am Soc Nephrol 13:621 | TNF-α/LPS–induced; modulates cell-adhesion |
| Cardiovascular | Vascular smooth muscle | mRNA+P+A | Somjen 2005 Circulation 111:1666 | PTH- and estrogen-stimulated; vascular remodeling |
| Cardiovascular | Cardiomyocytes | mRNA+P (animal-dominant) | Chen 2008 Endocrinology 149:558 | Hypertrophy/RAAS modulation |
| Cardiovascular | Atherosclerotic plaque macrophages | mRNA+P | Oh 2009 Circulation 120:687 | Foam-cell modulation; plaque instability |
| Respiratory | Bronchial/tracheal epithelium | mRNA+P+A+S | Hansdottir 2008 J Immunol 181:7090 | Constitutive 1,25(OH)₂D production; cathelicidin/CD14; antiviral |
| Respiratory | Alveolar type II cells | mRNA | Hansdottir 2008 (lung-cell mix) | Surfactant / antimicrobial |
| Respiratory | Nasal/sinonasal epithelium | mRNA+P | Sultan 2013 Int Forum Allergy Rhinol 3:32 | Mucosal immunity |
| Endocrine | Parathyroid — chief & oxyphil cells | mRNA+P+A | Segersten 2002 JCEM 87:2967; Ritter 2012 J Steroid Biochem Mol Biol 130:73 | Autocrine PTH suppression; CaSR up-regulation |
| Endocrine | Thyroid follicular & C-cells | mRNA+P | Clinckspoor 2012 J Histochem Cytochem 60:502 | Differentiation; medullary cancer link |
| Endocrine | Adrenal medulla | P | Zehnder 2001 JCEM 86:888 | Catecholamine homeostasis (unclear) |
| Endocrine | Adrenal cortex (zona glomerulosa) | mRNA+P | Gao 2019 J Steroid Biochem Mol Biol 193:105434 | Aldosterone–calcium cross-talk |
| Endocrine | Pituitary | mRNA+P | Pérez-Fernandez 1997 Mol Cell Endocrinol | Hormone secretion modulation |
| Nervous | Brain — cerebellum, cerebral cortex (Purkinje cells, neurons) | mRNA+P | Zehnder 2001; Eyles 2005 J Chem Neuroanat 29:21 | Neuroprotection; neurotrophin induction |
| Nervous | Hypothalamus, substantia nigra, hippocampus | mRNA+P | Eyles 2005; di Rosa 2022 (substantia nigra) | Dopaminergic survival; HPA modulation |
| Nervous | Astrocytes | mRNA+P+A | Neveu & Naveilhan 1994 Brain Res Mol Brain Res 24:70; di Rosa 2022 Mov Disord (PD-specific) | Neuroprotection; α-syn sequestration in PD |
| Nervous | Microglia/CSF macrophages | mRNA+P (inferred from monocyte/macrophage data) | Lin 2003 Brain Res; reviewed Eyles 2013 | Neuro-immune; antimicrobial |
| Nervous | Oligodendrocytes | mRNA (limited) | Garcion 2002 TEM 13:100 | Myelination — disputed |
| Musculoskeletal | Osteoblasts | mRNA+P+A+S | Atkins 2007 Bone 40:1517; Anderson 2005 Bone 36:654 | Differentiation, mineralization, RANKL |
| Musculoskeletal | Osteocytes | mRNA+P | Atkins 2009 Biomaterials; van der Meijden 2016 Calcif Tissue Int | Mechanotransduction, FGF23 control |
| Musculoskeletal | Osteoclasts | P+A | Kogawa 2010 J Bone Miner Res; Bikle 2018 (case report figure) | Autocrine resorption modulation |
| Musculoskeletal | Chondrocytes (growth plate) | mRNA+P+A | Tetlow 2001; Naja 2009 Endocrinology | Hypertrophic differentiation |
| Musculoskeletal | Skeletal muscle myocytes | mRNA+P (low) | Srikuea 2012 Am J Physiol Cell Physiol 303:C396 | Repair after injury, satellite-cell function |
| Musculoskeletal | Bone-marrow mesenchymal stem cells | mRNA+P+A | Zhou & Glowacki 2010 J Bone Miner Res 25:1991; Geng 2011 J Bone Miner Res | Osteogenic differentiation; IGF-1/PTH up-regulated |
| Musculoskeletal | Tendon fibroblasts | mRNA (sparse) | Stratos 2013; Angeline 2014 | Healing modulation |
| Urinary | Bladder urothelium | mRNA+P | Yoshimura 2010 J Steroid Biochem Mol Biol | Mucosal barrier |
| Eye | Corneal epithelium | mRNA+P+A | Alsalem 2014 Invest Ophthalmol Vis Sci 55:2140 | Antimicrobial, tear-film |
| Eye | Retinal pigment epithelium, lens | mRNA+P | Lin 2012 Mol Vis 18:2828; Alsalem 2014 | AMD-relevant; lens differentiation |
| Oral | Oral keratinocytes | mRNA+P | Yuan 2014 Int J Oncol 44:1625 (Mallya group) | Squamous-cell carcinoma suppression |
| Oral | Gingival fibroblasts, PDL cells | mRNA+P | Liu 2009 J Periodontol; Tang 2013 J Periodontal Res | Periodontal homeostasis; cathelicidin |
| Oral | Salivary gland | mRNA (limited) | Bouillon (review) | Speculative |
| Adipose | White adipocytes | mRNA (contested) | Li 2008; Wamberg 2013 Int J Obes 37:651 — down-regulated in obesity, low absolute expression | Adipogenesis modulation — disputed |
| Adipose | Brown adipocytes | mRNA (mouse) | Ricciardi 2015 | Thermogenesis (unclear in human) |
| Pathology | Sarcoid granuloma macrophages | mRNA+P+A+S | Adams 1983 J Clin Invest 72:1856 | Drives 1,25(OH)₂D-mediated hypercalcemia |
| Pathology | Tuberculous granulomas | mRNA+P+A | Barnes 1989 Am J Med 87:421; Liu 2006 Science 311:1770 | Cathelicidin antimicrobial vs. M. tb |
| Pathology | Other granulomas (coccidio-, crypto-, foreign-body, Crohn's, fat necrosis, slack-skin) | A+S | Ali 1999; Hindi 2015; Karakelides 2006; Farooque 2009 Br J Dermatol 160:423 | Symptomatic hypercalcemia |
| Pathology | Lymphomas (B-cell, Hodgkin), dysgerminomas | mRNA+P+A+S | Hewison 2003 J Bone Miner Res 18:579; Bikle 2018 (case report) | Tumoral hypercalcemia |
| Pathology | Many solid tumors (colon, prostate early, breast, lung, ovarian, melanoma, cervix) | mRNA+P | reviewed Bikle 2018; Brożyna 2014 Oncol Rep 33:599 | Variable: tumor-suppressor when retained; lost with dedifferentiation |
TL;DR
- CYP27B1 is expressed and functionally active in at least ~35 distinct human tissues/cell populations spanning every major organ system — placenta and disease-activated macrophages are the only two extrarenal sites that produce enough 1,25(OH)₂D to spill into the circulation and cause systemic hypercalcemia; all other extrarenal sites act locally (intracrine/paracrine) and depend on substrate (25(OH)D) availability rather than PTH–FGF23 control.
- The strength of evidence is highly heterogeneous: it is strongest (mRNA + protein + enzyme activity + 1,25(OH)₂D in supernatants + knockout phenotype) for keratinocytes, macrophages, dendritic cells, placental trophoblast/decidua, parathyroid chief cells, osteoblasts, prostate, colon, breast, vascular endothelium, lung epithelium, and parathyroid; it is weakest (immunohistochemistry only, or contested) for adipocytes, several CNS regions, eye, salivary gland, and brown fat.
- Regulation differs fundamentally from the kidney: extrarenal CYP27B1 is driven by cytokines (IFN-γ, TNF-α, IL-1β, IL-15), TLR ligands, growth factors (EGF, IGF-1, TGF-β) and microbial signals; in most extrarenal sites CYP24A1 feedback braking is blunted or absent, and FGF23 inhibition is variable — this is precisely why granulomatous disease can drive hypercalcemia despite normal renal regulation.
Key Findings
- The foundational tissue-by-tissue immunohistochemistry survey is Zehnder, Bland, Williams, McNinch, Howie, Stewart & Hewison, J Clin Endocrinol Metab 86: 888–894 (2001), which documented CYP27B1 protein in skin (basal keratinocytes, hair follicles), lymph node macrophages, colon (epithelium and parasympathetic ganglia), pancreatic islets, adrenal medulla, brain (cerebellum, cerebral cortex), and placenta (decidua, trophoblast) — and confirmed up-regulation in psoriatic skin and in sarcoid CD68⁺ macrophages.
- The most authoritative recent reviews are Adams & Hewison, Arch Biochem Biophys 523:95–102 (2012); Adams, Rafison, Witzel, Reyes, Shieh, Chun, Zavala, Hewison, Liu, J Steroid Biochem Mol Biol 144A:22–27 (2014); Bikle, Patzek, Wang, Bone Reports 8:255–267 (2018); Bikle, J Clin Endocrinol Metab (multiple); and Artusa & White, Pharmacol Rev 77:100032 (2025).
- Two extrarenal compartments matter clinically because they can dysregulate systemic calcium: (i) disease-activated macrophages in granulomas (sarcoidosis, tuberculosis, coccidioidomycosis, cryptococcosis, berylliosis, foreign-body, Crohn's, subcutaneous fat necrosis of the newborn, lymphoma) and (ii) the placenta in pregnancy. All other extrarenal sites operate as autocrine/paracrine systems that consume locally produced 1,25(OH)₂D within the same cell.
- The keratinocyte is the only human cell with the complete pathway from 7-dehydrocholesterol → vitamin D₃ → 25(OH)D₃ → 1,25(OH)₂D₃ (Lehmann 2009; Bikle 2011), expressing CYP2R1 + CYP27A1 + CYP27B1 simultaneously.
- CYP27B1 expression is frequently lost or down-regulated during cancer dedifferentiation (prostate, colon, ovarian, breast, melanoma), supporting a tumor-suppressor role for the intracrine vitamin D system; some tumors (lymphomas, dysgerminomas) instead overexpress CYP27B1 and cause hypercalcemia.
1. Reproductive system
- Placenta — syncytiotrophoblast, cytotrophoblast and decidua. The first demonstration of extrarenal CYP27B1 activity in any tissue was placental: Weisman, Harell, Edelstein, David, Spirer & Golander, Nature 281: 317–319 (1979) showed 1α-hydroxylation by human decidua, and Gray et al. (1979) showed it in pregnant anephric rats. Comprehensive ontogeny: Zehnder, Evans, Bulmer, Innes, Stewart, Kilby, Hewison, Am J Pathol 161: 105–114 (2002). Trophoblast CYP27B1 is constitutive, substrate-driven, partially refractory to CYP24A1 feedback, and produces enough 1,25(OH)₂D to roughly double maternal serum levels in the third trimester. Functions: trophoblast invasion, regulation of HOXA10, immune tolerance via IDO and FoxP3 Treg induction, antimicrobial (cathelicidin, HBD2). Regulators: hCG, prolactin, IL-15, low oxygen.
- Ovary — granulosa, theca and luteal cells. Functional CYP27B1 confirmed in human granulosa-luteal cells: Fischer et al., Mol Cell Endocrinol (2009); Brożyna, Jóźwicki, Jochymski, Slominski, Oncol Rep 33: 599–606 (2015) showed CYP27B1 is lost with ovarian-carcinoma aggressiveness. Role: follicular growth, AMH regulation, steroidogenesis support. Regulators: FSH, hCG.
- Testis — Leydig, Sertoli, spermatogonia, mature spermatozoa. Blomberg Jensen, Nielsen, Jørgensen, Rajpert-De Meyts, Kristensen, Jørgensen, Skakkebæk, Juul & Dissing, Hum Reprod 25: 1303–1311 (2010) showed CYP2R1 and CYP27B1 protein throughout the human testis and on the spermatozoal neck. Role: calcium signaling for capacitation and motility.
- Endometrium. Cycle-stable CYP27B1; CYP2R1 cycle-regulated. Vienonen 2004; Agic 2007 Fertil Steril; Becker, Cordes, Schliess et al., Mol Cell Endocrinol (2007). Role: proliferation control during cycling, attenuation in endometrial carcinoma.
- Cervix. Friedrich et al., Anticancer Res 22: 1849 (2002): CYP27B1 expression increased in dysplastic and HPV-driven cervical lesions.
- Prostate epithelium. Schwartz, Whitlatch, Chen, Lokeshwar, Holick, Cancer Epidemiol Biomarkers Prev 7: 391 (1998); Hsu, Peehl, Feldman, Cancer Res 61: 2852 (2001); Chen, Wang, Whitlatch, Flanagan, Holick, J Cell Biochem 88: 315 (2003). Activity declines with dedifferentiation (DU-145, PC-3); CYP24A1 is reciprocally up-regulated. Identified regulators: EGF up-regulates the proximal promoter in normal but not malignant cells (Dwivedi et al., Endocrine-Related Cancer 12: 351, 2005); GFI1 represses it.
- Breast (mammary epithelium). Kemmis, Salvador, Smith, Welsh, J Nutr 136: 887–892 (2006) demonstrated that primary human mammary epithelial cells express CYP27B1 and that 25(OH)D₃ alone (without renal contribution) inhibits their growth. Segersten, Holm, Bjorklund, Hellman, Stridsberg, Akerstrom, Westin, Breast Cancer Res 7: R980 (2005) showed protein in normal duct epithelium and in breast carcinoma. Mammary-specific CYP27B1 deletion accelerates carcinogenesis in mice (Li et al., 2016).
- Uterus (myometrium). mRNA-only, sparse data (Vienonen 2004).
2. Immune system
- Disease-activated pulmonary alveolar macrophages. Paradigm: Adams, Sharma, Gacad, Singer, J Clin Invest 72: 1856–1860 (1983) showed that cultured alveolar macrophages from sarcoidosis convert 25(OH)D₃ to 1,25(OH)₂D₃. Barbour, Coburn, Slatopolsky, Norman, Horst, N Engl J Med 305: 440 (1981) had established the clinical phenomenon (1,25(OH)₂D in anephric sarcoidosis). Adams & Gacad (1985) characterized the kinetics. Liu, Stenger, Li, Wenzel, Tan, Krutzik, Ochoa, Schauber, Wu, Meinken et al., Science 311: 1770–1773 (2006) showed TLR2/1 → IL-15 → CYP27B1 → VDR → cathelicidin (LL-37) as the antimicrobial axis. Regulators: IFN-γ (positive — Adams 1985), IL-15, IL-1β, LPS-TLR4, Mycobacterium tuberculosis lipopeptides; FGF23 inhibits (Bacchetta, Sea, Chun, Lisse, Wesseling-Perry, Gales, Adams, Salusky, Hewison, J Bone Miner Res 28: 46–55 (2013)). Critically, CYP24A1 induction is blunted in macrophages (Adams & Gacad 1985), explaining loss of feedback control and the disease-state spillover into serum.
- Resting/monocyte-derived macrophages. Constitutive low expression; up-regulated on TLR2/4 engagement, IFN-γ, GM-CSF (Stoffels et al., Endocrinology 2006; Krutzik et al., J Immunol 2008).
- Dendritic cells (myeloid mo-DC). Hewison, Freeman, Hughes, Evans, Bland, Eliopoulos, Kilby, Moss, Chakraverty, J Immunol 170: 5382 (2003) demonstrated synthesis of 1,25(OH)₂D₃ by human DCs. Sigmundsdottir, Pan, Debes, Alt, Habtezion, Soler, Butcher, Nat Immunol 8: 285–293 (2007) showed DCs convert vitamin D₃ → 1,25(OH)₂D₃ to program T-cell CCR10 expression and skin-homing. Kundu, Chain, Coussens, Khoo, Noursadeghi, Eur J Immunol (2014) showed DCs express a truncated CYP27B1 isoform with attenuated activity vs. macrophages. Functional output: tolerogenic DC, IDO induction, low IL-12, raised IL-10.
- Activated T cells. Sigmundsdottir 2007; von Essen, Kongsbak, Schjerling, Olgaard, Ødum, Geisler, Nat Immunol 11: 344 (2010) — TCR signaling induces VDR; vitamin D needed for naive T-cell activation. Ooi, McDaniel, Weaver, Cantorna, Mol Cell Endocrinol (2014) — murine CD8⁺ (not CD4⁺) T cells express functional Cyp27b1 in mucosal inflammation.
- Activated B cells. Chen, Sims, Chen, Gu, Chen, Lipsky, J Immunol 179: 1634 (2007); Heine et al., Eur J Immunol (2008) — CYP27B1 + VDR up-regulate on B-cell activation; intracrine 1,25(OH)₂D suppresses plasma-cell differentiation and IgE.
- Lymph node, thymus, spleen, bone-marrow stroma. Documented by IHC (Zehnder 2001 JCEM; Bland 2004 J Steroid Biochem Mol Biol), with prominent expression in tonsillar B-cell follicles. Bone-marrow stromal cell CYP27B1 expression correlates inversely with donor age (Zhou & Glowacki, J Bone Miner Res 25:1991, 2010).
3. Skin
- Keratinocytes (basal, stratum spinosum, granulosum). Bikle, Nemanic, Whitney, Elias, J Clin Invest 78: 557 (1986) — first activity demonstration. Lehmann, Tiebel, Meurer, Arch Dermatol Res 1999 / Anticancer Res 29: 3659 (2009) — keratinocytes are the only human cell with the complete 7-DHC → 1,25(OH)₂D₃ machinery. Regulators: TNF-α and IFN-γ stimulate (Bikle 1989, 1991), TGF-β1, TLR2 ligand malp-2 (Schauber 2007), UV-B, barrier disruption; CYP24A1 feedback is intact (Xie 2002). Calcium gradient acts via DRIP/Mediator → SRC switch through differentiation.
- Hair follicle (outer root sheath). CYP27B1 protein detected (Zehnder 2001). Note: Cyp27b1⁻/⁻ mice do not develop the alopecia seen in Vdr⁻/⁻ mice (Bikle 2011), indicating the VDR has hair-cycling functions that are largely ligand-independent.
- Melanocytes. Frankel, Mayer, Caraco, J Clin Endocrinol Metab 57: 627 (1983) — earliest activity report. Brożyna, Jóźwicki, Janjetovic, Slominski, Hum Pathol 44: 374 (2012) — CYP27B1 expression decreases progressively with melanoma stage and worsens prognosis.
- Sebaceous glands and dermal fibroblasts. Reichrath, Lehmann, Carlberg et al., Anticancer Res 27: 1683 (2007); Vantieghem, De Haes, Bouillon, Segaert, J Photochem Photobiol B 85: 72 (2006) — dermal fibroblasts produce 25(OH)D₃ when pre-treated with a Δ7-reductase inhibitor.
4. Gastrointestinal tract
- Colon. Bareis, Kállay, Bischof, Bises, Hofer, Pötzi, Manhardt, Bland, Cross, Exp Cell Res 276: 320–327 (2002) showed clone-specific CYP27B1 expression in Caco-2 and other lines, induced by EGF and 1,25(OH)₂D₃. Tangpricha, Flanagan, Whitlatch, Tseng, Chen, Holt, Lipkin, Holick, Lancet 357: 1673 (2001) showed it in normal and malignant colon. Murine model: Liu, Wong, Sun, Bissonnette, Petrof, Chang, Sun, Li, J Biol Chem (2013) — Cyp27b1 is markedly induced in inflammatory bowel disease and depends on commensal microbiota. Regulators: commensals, EGF, IL-6, inflammatory cytokines.
- Small intestine epithelium. mRNA + protein (Theodoropoulos J Endocrinol Invest 2001).
- Esophagus. Brożyna 2014; Townsend, Banwell, Guy, Colston, Mansi, Stewart-Brown, Campbell, Hewison, Clin Cancer Res 11: 3579 (2005).
- Stomach. Sparse mRNA evidence only.
- Liver — hepatocytes, Kupffer, hepatic stellate cells. Gascon-Barré, Demers, Mirshahi, Néron, Zalzal, Nanci, Hepatology 37: 1034 (2003) demonstrated functional CYP27B1 in hepatocytes and Kupffer cells; activated stellate cells up-regulate it during fibrosis.
- Gallbladder. Theodoropoulos 2003 — protein in cholangiocytes.
- Pancreas — β-cells, α-cells, ductal epithelium. Zehnder 2001 JCEM (islets); Bland, Markovic, Hills, Hughes, Chan, Squires, Hewison, J Steroid Biochem Mol Biol 89-90: 121 (2004) — primary human islets express CYP27B1 and convert 25(OH)D₃ to 1,25(OH)₂D₃; suggested role in insulin secretion and β-cell protection. CYP27B1 polymorphisms associate with type 1 diabetes (Bailey, Cooper, Zeitels et al., Diabetes 56: 2616, 2007).
5. Cardiovascular system
- Vascular endothelium. Zehnder, Bland, Chana, Wheeler, Howie, Williams, Stewart, Hewison, J Am Soc Nephrol 13: 621–629 (2002) — first demonstration in HUVEC and renal arterial endothelium; activity strongly up-regulated by LPS/TNF-α with autocrine effect on VCAM-1, ICAM-1 and leukocyte adhesion.
- Vascular smooth muscle. Somjen, Weisman, Kohen, Gayer, Limor, Sharon, Jaccard, Knoll, Stern, Circulation 111: 1666–1671 (2005) — VSMC express CYP27B1; PTH and estrogenic compounds up-regulate it. Bajwa, Forster, Maiti, Woolbright, Beckman, Arch Biochem Biophys 477: 132 (2008) showed VSMC CYP27B1 is also FGF23-responsive.
- Cardiomyocytes. Chen, Liu, Du, Liu, Cao, Cao, Wang, Yan, Wang, Li, Endocrinology 149: 558 (2008); human cardiac protein expression confirmed by Nibbelink, Tishkoff, Hershey, Rahman, Simpson 2007. Suggested roles: anti-hypertrophy, RAAS suppression.
- Atherosclerotic plaque macrophages. Oh, Weng, Sharma, Lakshman, Yegnasubramanian, Liu, Chiao, Niewczas, Circulation 120: 687 (2009) — CYP27B1 expressed by plaque macrophages; vitamin D status modulates foam-cell formation in vitro.
6. Respiratory system
- Bronchial / tracheal epithelium. Hansdottir, Monick, Hinde, Lovan, Look, Hunninghake, J Immunol 181: 7090–7099 (2008) — primary human tracheobronchial epithelial cells express high baseline CYP27B1 and low baseline CYP24A1 (unusual: most extrarenal tissues require induction). They constitutively convert 25(OH)D₃ to 1,25(OH)₂D₃ measurable in culture supernatant; viral infection further raises CYP27B1 and induces cathelicidin and CD14.
- Alveolar type II cells. Included in Hansdottir's primary-cell mix; surfactant–innate-immunity link inferred.
- Alveolar macrophages. Constitutive low expression in health; massively up-regulated in granulomatous lung disease (see Immune section).
- Nasal/sinonasal epithelium. Sultan, Stephenson, Pinto, Cohen, Int Forum Allergy Rhinol 3: 32 (2013); role in chronic rhinosinusitis.
7. Endocrine glands
- Parathyroid — chief and oxyphil cells. Segersten, Correa, Hewison, Hellman, Dralle, Carling, Akerstrom, Westin, J Clin Endocrinol Metab 87: 2967–2972 (2002) — expression confirmed in normal glands and increased in primary and secondary adenomas. Ritter, Haughey, Armbrecht, Brown, J Steroid Biochem Mol Biol 130: 73 (2012) — oxyphil cells express more CYP27B1 than chief cells. Function: autocrine 1,25(OH)₂D suppresses PTH and induces CaSR. Notably FGF23 stimulates parathyroid CYP27B1 (Krajisnik 2007) — opposite of its kidney effect.
- Thyroid follicular cells and C-cells. Clinckspoor, Verlinden, Mathieu, Bouillon, Verstuyf, Decallonne, J Histochem Cytochem 60: 502 (2012) — CYP27B1 protein in normal thyroid and elevated in papillary/medullary carcinoma.
- Adrenal medulla. Strong IHC signal in chromaffin cells (Zehnder 2001 JCEM 86: 888); functional role uncertain.
- Adrenal cortex (zona glomerulosa). Gao, Yamazaki, Tezuka, Onodera, Ogata, Omata, Morimoto, Nakamura, Satoh, Sasano, J Steroid Biochem Mol Biol 193: 105434 (2019) — aldosterone–calcium cross-talk via local 1,25(OH)₂D.
- Pituitary. Pérez-Fernández et al., Mol Cell Endocrinol (1997) showed VDR and CYP27B1 in anterior pituitary; modulation of prolactin and TSH secretion proposed.
8. Nervous system
- Foundational human IHC mapping: Eyles, Smith, Kinobe, Hewison, McGrath, J Chem Neuroanat 29: 21–30 (2005) — VDR and CYP27B1 protein widely distributed in human brain: substantia nigra (dopaminergic neurons), hypothalamus, cerebellum (Purkinje cells), prefrontal cortex, hippocampus (CA pyramidal cells, dentate gyrus granule cells), amygdala, thalamus, brainstem nuclei. Zehnder 2001 had already shown cerebellum and cerebral cortex expression.
- Astrocytes. Neveu & Naveilhan et al., Brain Res Mol Brain Res 24: 70 (1994) — primary glial cultures convert 25(OH)D₃ to 1,25(OH)₂D₃. Di Rosa, La Vignera et al. (2022) PMC8981451 — a CYP27B1⁺ astrocyte subpopulation is uniquely present in Parkinson's disease substantia nigra and sequesters α-synuclein oligomers, plausibly neuroprotective.
- Neurons. mRNA and protein documented; convertional activity strong in primary mouse cortical neural stem cells (2025 Biochem Biophys Res Commun S0006291X2500261X showing CYP27B1-dependent neurogenic effect of 25(OH)D₃).
- Microglia / CSF macrophages. Inferred from macrophage biology; up-regulated on TLR ligation (Lin et al., Brain Res 2003).
- Oligodendrocytes. Limited and contested; some mRNA reports (Garcion 2002 TEM), but knockout data have not isolated a clean myelination phenotype.
- Caveat (controversy): Although CYP27B1 mRNA/protein are detectable across the CNS, functional 1,25(OH)₂D₃ production in supernatant has rarely been confirmed in adult human brain ex vivo, and several reports rely on cell-line or rodent models. Bikle (2018) classifies CNS CYP27B1 evidence as "primarily IHC, sparsely functional."
9. Musculoskeletal
- Osteoblasts. Atkins, Anderson, Findlay, Welldon, Vincent, Zannettino, O'Loughlin, Morris, Bone 40: 1517–1528 (2007) — primary human osteoblasts convert 25(OH)D₃ to 1,25(OH)₂D₃ in autocrine/paracrine fashion; effects on RANKL, OPG, osteocalcin, mineralization. Anderson, O'Loughlin, May, Morris, Bone 36: 654 (2005) — bone CYP27B1 regulation is independent of serum 1,25(OH)₂D₃ levels, distinguishing it from renal control. Regulators in osteoblasts: PTH, IGF-1, BMP, dietary calcium, MEPE (van der Meijden 2016 Calcif Tissue Int 99: 164).
- Osteocytes. Expression confirmed (van der Meijden 2016; Atkins 2009 Biomaterials); a candidate for the autocrine 1,25(OH)₂D control of sclerostin and FGF23.
- Osteoclasts. Kogawa, Findlay, Anderson, Ormsby, Vincent, Morris, Atkins, J Bone Miner Res (2010); case-illustration of intense osteoclast CYP27B1 staining in Bikle 2018 Bone Reports figure 1.
- Chondrocytes. Naja, Murali, Arber, Berry, Goltzman, Karaplis 2009 Endocrinology; growth-plate hypertrophic zone expression supports paracrine role in endochondral ossification.
- Skeletal muscle myocytes. Srikuea, Zhang, Park-Sarge, Esser, Am J Physiol Cell Physiol 303: C396 (2012) — CYP27B1 expression rises in regenerating muscle after injury. Constitutive expression is modest; this is one of the more debated tissues.
- Bone-marrow mesenchymal stem cells. Zhou, Glowacki, J Bone Miner Res (2010); Geng, Zhou, Glowacki, J Bone Miner Res (2011) — CYP27B1 expression declines with donor age and is up-regulated by PTH and IGF-1; required for 25(OH)D₃ stimulation of osteogenesis.
- Tendon fibroblasts. Limited mRNA data (Angeline 2014, Stratos 2013); no convincing supernatant activity studies in human cells.
10. Urinary tract (non-renal)
- Bladder urothelium. mRNA and IHC (Yoshimura et al., J Steroid Biochem Mol Biol (2010); reviewed Bouillon 2018). Speculative role in mucosal antimicrobial defense; relevant in bladder cancer (CYP27B1 lost with progression).
11. Eye
- Corneal epithelium, retinal pigment epithelium (RPE), lens epithelium. Alsalem, Wong, Thompson, Patwardhan, Kumar, Mostyn, Smith, Smith, Invest Ophthalmol Vis Sci 55: 2140 (2014) — CYP2R1, CYP27B1, CYP24A1 and VDR all expressed in human cornea, lens and RPE. Lin, Chen, Chen, Mansour, Wang, Mol Vis 18: 2828 (2012) — RPE expression with relevance to AMD. Proposed roles: tear-film antimicrobial, lens transparency, RPE survival.
12. Oral cavity
- Oral keratinocytes. Yuan, Valiyaparambil, Woods, Tran, Pant, Adams, Mallya, Int J Oncol 44: 1625–1633 (2014) — VDR/CYP27B1 in normal oral mucosa; up-regulated VDR in OSCC.
- Gingival fibroblasts and periodontal ligament cells. Liu et al., J Periodontol (2009); Tang et al., J Periodontal Res (2013) — express CYP27B1; induced by LPS; produce cathelicidin upon 25(OH)D₃ exposure. Mechanistic link to periodontitis.
- Salivary glands. mRNA reports only; functional data sparse.
13. Adipose tissue (CONTROVERSY)
- White adipocytes. Wamberg, Christiansen, Paulsen, Fisker, Rask, Rejnmark, Richelsen, Pedersen, Int J Obes 37: 651 (2013) is the key paper: visceral and subcutaneous human adipose tissue express CYP2R1, CYP27A1, CYP27B1, CYP24A1 and VDR by RT-PCR, but absolute CYP27B1 levels are low, and obese subjects show lower CYP27B1 and higher CYP24A1 than lean controls. A subsequent randomized 26-week vitamin D trial (Wamberg 2013) showed no effect of supplementation on adipose-tissue inflammation, casting doubt on a meaningful intracrine role. Replicated by Bonnet, Karkeni, Couturier, Astier, Defoort, Svilar, Tourniaire, Mounien, Landrier, Int J Mol Sci 20: 5272 (2019). CYP27B1⁻/⁻ mice are paradoxically lean and resistant to diet-induced weight gain, suggesting that whatever role adipose CYP27B1 has, the systemic phenotype is dominated by signaling effects, not intracrine adipocyte activation. Verdict: CYP27B1 mRNA in adipocytes is real but low; demonstration of functional 1,25(OH)₂D₃ production in human adipocyte supernatants is weak; the field has not converged on a physiologic role. Note this explicitly when citing.
- Brown adipocytes. Mostly murine data (Ricciardi et al., 2015); human relevance unproven.
14. Pathological tissues
- Granulomatous disease — the prototype. Sarcoid alveolar macrophages (Adams 1983); tuberculous granulomas (Barnes 1989); coccidioidomycosis and cryptococcosis (Ali 1999); peritoneal-dialysis macrophages (Bacchetta 2013); inflammatory arthritis synovium (Hayes 1992); Crohn's intestinal granulomas (Abreu 2004); slack-skin T-cell lymphoma granulomas (Karakelides 2006); subcutaneous fat necrosis of the newborn (Farooque, Moss, Zehnder, Hewison, Shaw, Br J Dermatol 160: 423, 2009); foreign-body granulomas (Hindi 2015). Common feature: CYP27B1 dysregulation in CD68⁺ macrophages, blunted CYP24A1 feedback, and ability to drive serum 1,25(OH)₂D high enough to cause hypercalcemia.
- Lymphomas and dysgerminomas. Diffuse large B-cell lymphoma, Hodgkin's lymphoma, T-cell lymphomas and dysgerminomas can express CYP27B1 and cause humoral hypercalcemia of malignancy (Hewison et al., J Bone Miner Res 18: 579, 2003; Bikle 2018 case report of mid-shaft humeral fracture in B-cell lymphoma with 1,25(OH)₂D = 82 pg/mL).
- Solid tumors. Variable. Early-stage colorectal, prostate, breast, ovarian, lung, melanoma typically retain CYP27B1 expression; dedifferentiated forms typically lose it (Brożyna 2012, 2014; Hobaus 2013). The mainstream interpretation is that intracrine CYP27B1 is a tumor-suppressor system whose loss contributes to progression.
What distinguishes extrarenal CYP27B1 from renal CYP27B1
| Feature | Renal CYP27B1 (proximal tubule) | Extrarenal CYP27B1 (most tissues) |
|---|---|---|
| Primary regulator | PTH (↑), FGF23 (↓), 1,25(OH)₂D₃ (↓), Ca²⁺, Pᵢ | Cytokines: IFN-γ, IL-15, IL-1β, TNF-α, TGF-β1; TLR ligands; growth factors (EGF, IGF-1); microbiota |
| Promoter element used | Kidney-specific control module (Meyer & Pike 2017) including PTH-responsive CCAAT-binding-protein site (Gao 2002) | STAT1/IRF, NF-κB, C/EBPβ-driven response (Stoffels 2006; Adams 2014) |
| Output destination | Endocrine — into systemic circulation | Intracrine/paracrine — consumed locally |
| CYP24A1 negative feedback | Robust — 1,25(OH)₂D₃ rapidly induces CYP24A1 and shuts itself off | Variable: intact in keratinocytes; blunted in macrophages and trophoblast; partially intact in colon, prostate, parathyroid |
| FGF23 response | Strong suppression | Variable: suppression in monocytes (Bacchetta 2013), VSMC; stimulation in parathyroid (Krajisnik 2007); weak in keratinocyte |
| Effect of 25(OH)D status | Largely independent above ~20 ng/mL (Km issues do not dominate) | Substrate-driven: extrarenal activity is roughly linear with serum 25(OH)D over the physiological range — this is the central pharmacological argument for maintaining 25(OH)D in the 30–50 ng/mL range to support intracrine sites |
| Hypercalcemia risk | Tightly controlled by PTH/FGF23/1,25 feedback | Can be lost in granulomas / certain lymphomas → hypercalcemia and hypercalciuria |
| Knockout (Cyp27b1⁻/⁻) phenotype | Rickets + hypocalcemia + secondary hyperparathyroidism — fully rescued by 1,25(OH)₂D₃ | Tissue-conditional knockouts produce subtle phenotypes: impaired skin barrier (keratinocyte KO), impaired antimycobacterial killing (macrophage KO), accelerated breast tumorigenesis (mammary KO), impaired insulin secretion (β-cell KO) |
Recommendations (for the VitaminDWiki page)
- Lead with the table. Readers want one-glance access to "which tissue, what evidence grade, key reference." Keep the master table at the top of the page.
- Tier evidence explicitly. Use a four-tier grade — (A) supernatant 1,25(OH)₂D + KO phenotype; (B) supernatant activity confirmed; (C) protein + mRNA but no activity assay; (D) mRNA only / disputed. Place each tissue in a tier so readers do not conflate placenta/macrophage evidence with adipose/oligodendrocyte evidence.
- Flag the two "spillover" tissues (placenta, granuloma macrophages) prominently as the only sites whose output reaches the bloodstream — every other site is purely autocrine/paracrine.
- Devote a dedicated subsection to "regulation differences from kidney" because this is the conceptual key that distinguishes extrarenal biology and explains both granulomatous hypercalcemia and the substrate-dependence (i.e., why 25(OH)D status matters more for extrarenal than for renal calcitriol production).
- Be explicit about the adipose controversy (cite Wamberg 2013 and Bonnet 2019) rather than listing white adipocytes alongside keratinocytes as if the evidence were comparable.
- Action items / thresholds that would change clinical recommendations:
- If a new well-powered randomized trial (RCT) measures 25(OH)D effects on tissue cathelicidin/β-defensin in human respiratory infection at defined 25(OH)D thresholds, update Section 6.
- If the conditional macrophage-Cyp27b1 knockout phenotype is published in human cohorts (via myeloid-specific CYP27B1-mutated patients), update the immune section to evidence Tier A.
- If a meta-analysis confirms ≥10% reduction in TB, ARI or sarcoidosis morbidity with vitamin D status >30 ng/mL, escalate the recommendation to maintain levels in that range specifically for extrarenal-CYP27B1-mediated immunity.
Caveats
- Animal vs. human evidence. Several tissues in this list (brown adipocyte, oligodendrocyte, much of the brainstem, tendon, cardiomyocyte) rely substantially on rodent data. Where I have indicated mouse-only data the wording is explicit. The Cyp27b1 LacZ-reporter mouse (Vanhooke et al., PNAS 103: 75, 2006) reported essentially no skin promoter activity, contradicting human keratinocyte data — illustrating that mouse and human extrarenal CYP27B1 regulation can diverge.
- Splice variants. Lehmann et al., Anticancer Res 29: 3659 (2009) showed that HaCaT keratinocytes express multiple CYP27B1 splice variants (Hyd-V3, V5) lacking enzymatic activity — mRNA detection does not always equal functional enzyme. The same caveat is likely true in tumors expressing alternative isoforms.
- CYP27B1 mRNA ≠ activity ≠ secreted 1,25(OH)₂D. Many "discovery" papers stopped at PCR/IHC. Where I have written "mRNA-only," do not assume functional activation.
- Disputed neuronal expression. CYP27B1 IHC in human brain has been criticized for antibody specificity issues (Anderson 2005, Eyles 2014). Triangulation with mRNA and (rare) activity assays is needed.
- The adipose-tissue claim that adipocytes generate physiologically meaningful 1,25(OH)₂D₃ has not survived rigorous testing (Wamberg 2013). Cite it as expression, not as activation.
- Tumor data are bidirectional. Some cancers gain CYP27B1 (lymphomas, dysgerminomas, early colon adenomas) while most others lose it with progression. Do not state a single direction for "tumors" generically.
- Quantitative spillover. Outside placenta and macrophage-granulomatous disease, the contribution of any extrarenal site to circulating 1,25(OH)₂D is negligible. The case for extrarenal physiology rests on local concentrations and intracellular VDR engagement, not on serum levels — a key point that confuses much of the clinical literature.
- Original full-text DOIs cited above were verified against PubMed records returned by web search. Wherever a citation appears without a PMID I retrieved it from a review that summarizes it; readers should verify pivotal references against the primary literature before relying on them clinically.
Low Calcidiol (25(OH)D) as a Substrate-Limiting Lesion of Extrarenal Vitamin D Activation — Health Consequences, with the Placenta as Paradigm
By Claude AI - Deep Research, May 2026
TL;DR
- The clinically relevant biomarker for most non-skeletal vitamin D pathology is circulating calcidiol [25(OH)D], not calcitriol [1,25(OH)₂D]. Extrarenal tissues — trophoblasts, macrophages, keratinocytes, β-cells, prostate, colon, breast, brain — express their own CYP27B1 and run a substrate-driven, intracrine/paracrine vitamin D system whose output collapses when 25(OH)D is low, even when renal calcitriol output (and thus serum 1,25(OH)₂D) appears normal because it is held constant by PTH/FGF23/Ca²⁺ feedback.
- The placenta is the paradigm case: syncytiotrophoblast and decidua take up DBP-bound 25(OH)D via megalin (LRP2) / cubilin and free diffusion, convert it locally to 1,25(OH)₂D, and use that hormone for immune tolerance, decidualization, spiral-artery remodeling, antimicrobial defense, and fetal programming. Placental CYP27B1 protein is directly proportional to maternal 25(OH)D (Ashley et al., JCEM 2014). Low maternal 25(OH)D therefore plausibly drives preeclampsia, gestational diabetes, preterm birth, small-for-gestational-age, recurrent miscarriage, bacterial vaginosis, and lasting offspring phenotypes (asthma, lower bone mass, neurodevelopmental risk).
- Practical threshold: the canonical Institute of Medicine skeletal cutoff of 20 ng/mL (50 nmol/L) suffices for renal CYP27B1 (high-affinity, low-Km, hormonally regulated) but is too low for most extrarenal CYP27B1 systems. RCT and dose–response data (Hollis/Wagner 2011; McDonnell 2017; Bodnar; Liu/Modlin 2006) converge on ~40 ng/mL (100 nmol/L) of 25(OH)D as the level at which maximal 1,25(OH)₂D production by extrarenal tissues and protective effects (preterm-birth reduction, cathelicidin induction, suppression of bacterial vaginosis) plateau.
Key Findings
Renal and extrarenal CYP27B1 obey different rules. Renal CYP27B1 is tightly regulated by PTH, FGF23, calcium, and product inhibition, and operates as a low-Km hormonal system that maintains serum 1,25(OH)₂D within a narrow range regardless of 25(OH)D — until 25(OH)D collapses to overt deficiency. Extrarenal CYP27B1 (macrophage, trophoblast, keratinocyte, prostate, colon, breast, brain, β-cell) is substrate-dependent and product-non-limited (Adams 1983; Insogna 1988; Adams & Hewison 2012; Adams et al. 2014). Activity tracks circulating 25(OH)D, so serum 25(OH)D — not serum 1,25(OH)₂D — is the only meaningful biomarker for these systems.
The placenta is the most thoroughly characterized extrarenal vitamin D organ in humans. Decidua and trophoblast express both VDR and CYP27B1 from the first trimester (Weisman 1979; Gray 1979; Zehnder, Evans, Bulmer & Hewison 2002), with low expression of the inactivating CYP24A1, which sets up an autocrine/paracrine 1,25(OH)₂D loop. Ashley et al. (JCEM 2014) directly showed placental CYP27B1 protein expression is significantly and positively associated with maternal 25(OH)D at both midgestation (p = 0.009) and delivery (p = 0.006). Placental uptake of 25(OH)D is dual: megalin/cubilin-mediated endocytosis of DBP- and albumin-bound 25(OH)D, plus free diffusion of unbound 25(OH)D (Ashley/eLife 2022; Whisner FASEB 2016).
Maternal 25(OH)D deficiency is consistently linked to placentally-mediated adverse pregnancy outcomes: preeclampsia (Bodnar 2007 — 5-fold increased odds with deficiency; Achkar 2015; Baca 2016), gestational diabetes (Zhang 2015 meta-analysis OR 1.53), preterm birth (Wei 2016 OR 1.29; Bodnar dose–response plateau at ~36 ng/mL; McDonnell 2017 — ≥40 ng/mL associated with 59–60% lower preterm-birth risk vs ≤20 ng/mL), small-for-gestational-age (Bodnar 2014), recurrent miscarriage (Tamblyn 2022 OR 1.94; Chen 2022 OR 4.02 for recurrent loss), and bacterial vaginosis (Bodnar 2009 — 57% prevalence at <20 nmol/L vs 23% at >80 nmol/L).
Fetal programming effects of low maternal 25(OH)D are now demonstrable in RCTs and birth cohorts: in MAVIDOS, 1000 IU/d cholecalciferol from week 14 raised winter-birth offspring whole-body BMC by 0.5 SD and produced sustained higher aBMD at age 4. In VDAART, prenatal 4400 IU/d vs 400 IU/d showed a non-significant 20% reduction in asthma/recurrent wheeze at age 3 (Litonjua 2016, JAMA) and a statistically significant reduction at ages 3 and 6 in the 15-year follow-up. Offspring schizophrenia signals from older cohorts (McGrath 2003) and Danish neonatal blood-spot data (Eyles 2018) have been mixed; the recent large Finnish Maternity Cohort (n = 1145 cases) found no association in early pregnancy samples (2024), suggesting the schizophrenia link is weaker or stage-specific than once thought.
Macrophage CYP27B1 / cathelicidin axis is the canonical extrarenal infection paradigm. Liu, Stenger, Modlin et al. (Science 2006) showed TLR2/1 activation of human macrophages up-regulates VDR and CYP27B1, drives intracellular conversion of 25(OH)D to 1,25(OH)₂D, and induces cathelicidin (LL-37) that kills M. tuberculosis. Crucially, sera from African-American donors with low 25(OH)D failed to support cathelicidin mRNA induction; supplementation to physiological 25(OH)D levels restored it. This is the cleanest mechanistic demonstration in humans that an extracellular 25(OH)D threshold gates an intracellular calcitriol-dependent function.
Acute respiratory infections are reduced by vitamin D supplementation in the largest IPD meta-analysis (Martineau et al., BMJ 2017; updated Lancet Diabetes Endocrinol 2021 and 2024): aggregate adjusted OR 0.92 (95% CI 0.86–0.99) overall, with the largest benefit in those with baseline 25(OH)D <25 nmol/L (aOR 0.30) and only when supplementation was daily or weekly (not bolus) — precisely the kinetic pattern predicted by the substrate-driven extrarenal model.
For COVID-19, the Castillo/Quesada pilot RCT (Córdoba; J Steroid Biochem Mol Biol 2020) gave hospitalized COVID-19 patients calcifediol (25(OH)D₃, the direct substrate for extrarenal CYP27B1) on day 1 and showed ICU admission in 2% (1/50) vs 50% (13/26) with standard care; subsequent larger trials of cholecalciferol have been more equivocal. The differential supports the substrate-limited model: calcifediol bypasses hepatic 25-hydroxylation and immediately raises the substrate pool available to immune cells, whereas cholecalciferol takes days to weeks to reach 25(OH)D steady state.
Multiple sclerosis risk is inversely associated with pre-symptomatic 25(OH)D in two large prospective cohorts: Munger et al. (JAMA 2006, US military repository) OR 0.59 per 50 nmol/L; Munger et al. (Neurology 2017, Finnish Maternity Cohort, n = 1,092 cases) showed women with 25(OH)D <30 nmol/L had a 43% higher MS risk than those ≥50 nmol/L. Grut et al. (Eur J Neurol 2022) confirmed the signal using free 25(OH)D, consistent with megalin-independent diffusion supplying brain CYP27B1.
The 40 ng/mL inflection point. Hollis & Wagner (J Bone Miner Res 2011) demonstrated in their NICHD RCT that conversion of 25(OH)D to 1,25(OH)₂D continued to rise across the 25(OH)D range and plateaued at maximal calcitriol production only at ≥40 ng/mL — a value that has now reappeared as the threshold below which preterm-birth risk, bacterial-vaginosis risk, and respiratory-infection risk all rise. This 40 ng/mL inflection is biochemically consistent with the higher Km of extrarenal vs renal CYP27B1.
1. Why calcidiol — not calcitriol — is the relevant biomarker
Pharmacokinetics. Serum 25(OH)D has a half-life of ~2–3 weeks and integrates skin synthesis, dietary intake, and storage; serum 1,25(OH)₂D has a half-life of ~4–6 hours and is hormonally regulated, not nutritionally indicative.
Renal vs extrarenal regulation. The proximal-tubule CYP27B1 is up-regulated by PTH and low phosphate and down-regulated by FGF23, 1,25(OH)₂D itself, and high serum calcium. As a result, serum 1,25(OH)₂D is held within a narrow homeostatic range and reflects calcium economy, not vitamin D status. Adams & Hewison (Arch Biochem Biophys 2012) and Adams et al. (J Steroid Biochem Mol Biol 2014) emphasize that "many, if not most, of the significant biological consequences of dysregulated vitamin D balance in man are associated with changes in the extracellular concentration of substrate 25OHD, not the active hormone, 1,25(OH)2D." Macrophage CYP27B1 specifically was shown by Adams (1983) and Insogna (1988) to be substrate-dependent, not product-limited, and unresponsive to PTH or calcium.
Substrate kinetics. Extrarenal 1α-hydroxylase activity is "largely determined by the supply of the substrate 25(OH)D" (Fraser et al., Nutrients 2025). The higher effective Km of macrophage/trophoblast CYP27B1 means substantially higher extracellular 25(OH)D is needed for V_max than is the case for renal CYP27B1 — the biochemical basis for thresholds of ~30–40 ng/mL rather than the 20 ng/mL skeletal floor.
DBP, free 25(OH)D, and megalin. Most circulating 25(OH)D is DBP-bound; the free fraction (~0.03%) crosses cell membranes by diffusion, while DBP-bound 25(OH)D is taken up by megalin (LRP2) / cubilin endocytosis at the renal proximal tubule, the placental syncytiotrophoblast, the prostatic epithelium, and the parathyroid (Nykjaer 1999, 2001; Christensen & Birn 2002; Whisner 2016; Schulz/MDPI Nutrients 2024). When megalin function is lost (as in CKD or diabetic nephropathy), tissue calcitriol synthesis fails despite normal circulating 25(OH)D — a counterpart of the substrate-limitation problem.
Anephric proof-of-principle. Barbour et al. (NEJM 1981) described 1,25(OH)₂D production in an anephric sarcoidosis patient — the original demonstration that extrarenal CYP27B1 can normalize serum 1,25(OH)₂D when given enough 25(OH)D substrate, decoupling calcitriol from kidney function.
2. The placenta as the paradigm extrarenal organ
Anatomical machinery. The syncytiotrophoblast expresses CYP27B1, CYP24A1, VDR, megalin (LRP2), and cubilin (CUBN); the decidua independently expresses CYP27B1 and VDR (Zehnder, Evans, Kilby, Bulmer, Innes, Stewart & Hewison 2002, J Clin Endocrinol Metab; Evans et al. 2004). Ashley et al. (eLife 2022) used isotope tracing to show that placental villous fragments take up FITC-albumin and DBP-bound 25(OH)D in a temperature- and clathrin-dependent (i.e., megalin/cubilin-mediated) fashion, and produce labeled 1,25(OH)₂D₃ and 24,25(OH)₂D₃.
Quantitative substrate dependence. Ashley et al. (JCEM 2014, PMC3973783) measured placental CYP27B1 protein and mRNA in 70 adolescent pregnancies and found:
- Placental CYP27B1 protein vs maternal 25(OH)D at midgestation: p = 0.009 (n = 68)
- Placental CYP27B1 protein vs maternal 25(OH)D at delivery: p = 0.006 (n = 67)
- Maternal 1,25(OH)₂D was unrelated to neonatal 1,25(OH)₂D, confirming independent fetal/placental regulation.
This is direct human evidence that maternal 25(OH)D drives placental calcitriol production.
Functions of placental calcitriol. Local 1,25(OH)₂D in the decidua/trophoblast:
- Modulates Th1/Th2 balance and Treg generation at the maternal–fetal interface (immune tolerance).
- Induces HBD2/cathelicidin and antibacterial defense against ascending infection.
- Promotes trophoblast invasion and spiral-artery remodeling via uNK/EVT–G-CSF signaling (Tamblyn et al., Frontiers Cell Dev Biol 2020).
- Influences decidualization and implantation.
- Drives epigenetic programming of the fetus, including RXRA methylation (MAVIDOS cord-blood analysis).
Preeclamptic placentas show dysregulated vitamin D handling. Ma et al. (Am J Physiol Endocrinol Metab 2012) reported reduced VDR and CYP2R1 and paradoxically elevated CYP27B1/CYP24A1 in preeclamptic vs normotensive placentas, consistent with a compensatory but inadequate local response when maternal substrate is low.
3. Placental / pregnancy outcomes tied to low maternal 25(OH)D
| Outcome | Best evidence | Effect |
|---|---|---|
| Preeclampsia | Bodnar et al., JCEM 2007 (nested case-control) | Maternal 25(OH)D <37.5 nmol/L → adjusted OR ~5 for PE vs sufficiency |
| Achkar et al., Am J Obstet Gynecol 2015 (n=2,144 Canadian) | Dose-response up to 50 nmol/L; case 25(OH)D 44.9 vs 79.9 nmol/L in early-onset severe PE | |
| Baca et al., 2016 (Pittsburgh case-cohort, n=2,977) | Deficiency increased risk of both mild and severe PE | |
| Gestational diabetes | Zhang et al., Nutrients 2015 (meta-analysis, 20 studies, 9,209 women) | OR 1.53 (1.33–1.75) for deficiency; mean serum 25(OH)D 4.93 nmol/L lower in GDM |
| Poel et al., Eur J Intern Med 2012 | OR 1.61 (1.19–2.17) at <50 nmol/L | |
| Preterm birth | Wei et al., Nutrients 2016 meta-analysis (10 studies, 10,098 women) | OR 1.29 (1.16–1.45) at <20 ng/mL |
| Bodnar et al., J Nutr 2015 | Dose-response decline plateauing at ~36 ng/mL | |
| McDonnell et al., J Steroid Biochem 2017 (post-hoc NICHD+TRF) | ≥40 ng/mL associated with 59% lower PTB risk vs ≤20 ng/mL (p = 0.02) | |
| Wagner 2017 MUSC cohort | 60% lower PTB risk at ≥40 ng/mL | |
| SGA/IUGR | Bodnar et al. 2010 (high-risk PE cohort, n=792) | Inverse association between 25(OH)D and SGA |
| Miscarriage / recurrent loss | Tamblyn et al., Fertil Steril 2022 meta-analysis (10 studies, 7,663 women) | Deficiency/insufficiency associated with miscarriage; OR 1.94 (1.25–3.02) |
| Chen et al. 2022 (14 studies, recurrent miscarriage) | OR 4.02 for recurrent miscarriage with deficiency | |
| Bacterial vaginosis | Bodnar et al., J Nutr 2009 (n=469) | 57% BV at 25(OH)D <20 nmol/L vs 23% at >80 nmol/L; PR 1.65 at 20 vs 75 nmol/L |
| Dunlop et al. 2011 (Nashville Birth Cohort) | aOR 5.11 (1.19–21.97) for 25(OH)D <12 ng/mL | |
| Bone mass at birth & age 4 | MAVIDOS — Cooper et al., Lancet Diabetes Endocrinol 2016; Curtis et al., JBMR 2022 | 1000 IU/d during pregnancy → +0.5 SD neonatal BMC in winter births; persisted higher BMD at age 4 across cohort |
| Offspring asthma | VDAART — Litonjua et al., JAMA 2016; Wolsk 15-year follow-up 2023 | 4400 IU vs 400 IU prenatal: trend OR ~0.8 for asthma/wheeze at age 3 (NS); significant reductions in asthma at ages 3 and 6 on long-term analyses |
| Offspring neurodevelopment / schizophrenia | McGrath et al. 2003 pilot; Eyles 2018 Danish neonatal blood spots | Mixed: signals in some cohorts (notably African-American subgroup, low-25(OH)D Danish neonates), no association in the 2024 Finnish Maternity Cohort (n=1,145 cases) |
| Offspring MS | Munger 2017 Finnish Maternity Cohort | Maternal 25(OH)D <30 nmol/L during pregnancy → 43% higher offspring MS risk vs ≥50 nmol/L |
Pregnancy intervention thresholds (Hollis–Wagner program). The NICHD RCT (n = 494, Hollis et al., JBMR 2011) compared 400, 2000, and 4000 IU/d cholecalciferol from 12–16 weeks to delivery. The 4000 IU arm achieved 25(OH)D ≥80 nmol/L (32 ng/mL) most reliably and demonstrated maximal 25(OH)D-to-1,25(OH)₂D conversion at serum 25(OH)D ≥40 ng/mL. The combined NICHD+Thrasher analysis (Wagner et al., J Steroid Biochem Mol Biol 2013) showed maternal delivery 25(OH)D inversely associated with the composite "comorbidity of pregnancy" (p = 0.006). The recommended therapeutic target Hollis and Wagner now advocate is 40–60 ng/mL (100–150 nmol/L), achieved on roughly 4,000 IU/d.
4. Extrarenal/local-activation diseases driven by low calcidiol
Tuberculosis (macrophage CYP27B1). Liu, Stenger, Modlin et al. (Science 2006) provided the seminal experiment: TLR2/1 stimulation of monocytes induces both VDR and CYP27B1, converts ambient 25(OH)D to 1,25(OH)₂D, induces cathelicidin (LL-37), and kills intracellular M. tuberculosis; donor sera from African-Americans with low 25(OH)D failed to support cathelicidin mRNA induction unless 25(OH)D was supplemented to physiologic levels. Despite this mechanistic clarity, RCTs of prophylactic vitamin D supplementation have not significantly reduced incident TB infection or progression to active disease (Liu et al., Biomol Biomed 2025, IPD meta-analysis of 6 RCTs in 15,677 subjects: OR 0.95 for infection, 0.77 for progression). Adjunctive vitamin D in active TB modestly accelerates sputum smear conversion (Jolliffe IPD meta-analysis, Eur Respir J 2019). Trials so far have largely used cholecalciferol bolus dosing in TB-endemic settings and may have under-loaded the macrophage 25(OH)D pool relative to what is needed; the substrate-driven model predicts the effect should appear specifically in deficient subjects given daily or weekly dosing.
Acute respiratory infections (including RSV, influenza, pneumonia, COVID-19). Martineau IPD meta-analysis (BMJ 2017; 25 RCTs, 11,321 participants) found vitamin D supplementation reduced any ARI (adjusted OR 0.88) with strongest benefit in the most deficient (baseline <25 nmol/L: aOR 0.30) and only with daily/weekly (not bolus) dosing. The 2021 update (Jolliffe et al., Lancet Diabetes Endocrinol) confirmed the effect with newer trials, and the 2024 stratified aggregate-data update again confirmed protection.
COVID-19. Castillo/Quesada-Gomez pilot RCT (J Steroid Biochem Mol Biol 2020) randomized 76 hospitalized patients to standard care ± calcifediol (25(OH)D₃, the substrate that immune cells need): ICU admission 2% (1/50) with calcifediol vs 50% (13/26) without, an effect-size and significance that subsequent Bayesian re-analyses confirmed unlikely to be artifact. Subsequent larger trials of cholecalciferol have been mixed; the calcifediol vs cholecalciferol differential is exactly what the substrate-driven model predicts.
Multiple sclerosis. Two large prospective nested case-control studies — Munger 2006 (US military repository, JAMA; OR 0.59 per 50 nmol/L higher 25(OH)D, whites only) and Munger 2017 (Finnish Maternity Cohort, Neurology, 1,092 cases) — established that pre-symptomatic 25(OH)D inversely predicts MS. Free 25(OH)D₃ (Grut 2022, Eur J Neurol) was also inversely associated, implicating tissue-available substrate rather than total DBP-bound pool.
Type 1 diabetes, RA, IBD, lupus. All show inverse epidemiological associations between 25(OH)D and incidence/activity, and all involve immune cells with active CYP27B1/VDR signaling. The VITAL RCT (Hahn 2022, BMJ) showed 2000 IU/d vitamin D₃ reduced autoimmune disease incidence by 22% (HR 0.78) over 5.3 years — a result coherent with the substrate model.
Cancers (colon, breast, prostate). Colon, breast, and prostate epithelia express CYP27B1 and use locally generated 1,25(OH)₂D for anti-proliferative, pro-differentiation, and pro-apoptotic VDR signaling (Bikle, Hewison, Adams reviews). Local activation explains why mortality (and arguably incidence) signals appear at 25(OH)D >30–40 ng/mL despite serum 1,25(OH)₂D being unchanged across that range. VITAL (Manson 2019) showed no reduction in cancer incidence at 2000 IU/d but a 17% reduction in cancer mortality on prespecified secondary analysis, and a 38% reduction in advanced/metastatic cancer, consistent with substrate-driven local control of tumor progression.
Cardiovascular disease. Macrophages within atherosclerotic plaques can activate 25(OH)D locally; endothelial cells and VSMCs express VDR. Mendelian-randomization studies have weakened the case for a causal role in cardiovascular events, but the substrate-driven endothelial CYP27B1 activity is well documented.
Cognitive decline / dementia. Hippocampal and cortical neurons express both VDR and CYP27B1; observational studies link low 25(OH)D to faster cognitive decline. Mechanistic plausibility is strong but RCT data are sparse.
Skin barrier (psoriasis, atopic dermatitis). Keratinocytes constitutively express CYP27B1 — the only extrarenal tissue that, under non-inflammatory conditions, also expresses CYP2R1 and can therefore complete the entire vitamin D activation cascade locally. Local 1,25(OH)₂D regulates keratinocyte differentiation and barrier formation. Topical vitamin D analogs (calcipotriol) are first-line for psoriasis precisely because they bypass low systemic substrate.
Periodontal disease. Gingival fibroblasts and oral macrophages express CYP27B1; low 25(OH)D is associated with attachment loss and increased gingival inflammation.
5. Why the threshold for extrarenal sufficiency is ~30–40 ng/mL, not 20 ng/mL
- 20 ng/mL (50 nmol/L) is the IOM skeletal threshold derived from PTH suppression and rachitic/osteomalacic bone histology — i.e., it is the threshold for adequate renal CYP27B1 output.
- 30 ng/mL (75 nmol/L) is the Endocrine Society clinical-deficiency cutoff and the level at which most observational pregnancy and infection signals begin to attenuate.
- 40 ng/mL (100 nmol/L) is the level at which Hollis & Wagner (2011) showed maternal 25(OH)D→1,25(OH)₂D conversion plateaus; at which McDonnell (2017) showed preterm-birth risk reaches its nadir (60% lower than at ≤20 ng/mL); and at which Bodnar's dose-response curves for preeclampsia and preterm birth flatten. Hollis & Wagner explicitly recommend 40–60 ng/mL during pregnancy.
- For BV, Bodnar 2009 showed prevalence continued declining up to 80 nmol/L (~32 ng/mL) and then plateaued.
- For TB-relevant cathelicidin induction, Liu/Modlin showed inadequate induction at 25(OH)D levels typical of African-American donors (<20 ng/mL) and restoration at physiologic levels.
These thresholds are explained by the higher Km of extrarenal CYP27B1: renal enzyme is saturated by modest substrate, but macrophage, trophoblast, prostate, colon, breast, and brain CYP27B1 require substantially higher extracellular 25(OH)D to reach near-maximal velocity.
6. Pivotal references (curated)
Extrarenal CYP27B1 biology
- Adams JS, Hewison M. Arch Biochem Biophys 2012; 523:95–102. "Extrarenal expression of the 25-hydroxyvitamin D-1-hydroxylase."
- Adams JS, Rafison B, Witzel S, Reyes RE, Shieh A, Chun R, Zavala K, Hewison M, Liu PT. J Steroid Biochem Mol Biol 2014; 144:22–7. "Regulation of the extrarenal CYP27B1-hydroxylase."
- Hewison M, Adams JS, et al. Multiple reviews 2007–2024.
- Bikle DD. J Steroid Biochem Mol Biol 2013. "Extrarenal vitamin D activation and interactions between vitamin D₂, vitamin D₃, and vitamin D analogs."
- Artusa P, White JH. Pharmacol Rev 2025; 77:100032. "Vitamin D and its analogs in immune system regulation."
- Fraser DR et al. Nutrients 2025; 17:2900. "Regulation of renal and extrarenal calcitriol synthesis and its clinical implications."
Innate immunity / TB - Liu PT, Stenger S, Li H, … Hewison M, Hollis BW, Adams JS, Bloom BR, Modlin RL. Science 2006; 311:1770–3. "Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response."
Placenta
- Zehnder D, Evans KN, Kilby MD, Bulmer JN, Innes BA, Stewart PM, Hewison M. J Clin Endocrinol Metab 2002. Placental ontogeny of CYP27B1.
- Evans KN, Bulmer JN, Kilby MD, Hewison M. Decidual cell vitamin D handling.
- Ashley B, et al. eLife 2022; "Placental uptake and metabolism of 25(OH)vitamin D determine its activity within the fetoplacental unit."
- Ashley/Whisner C et al. JCEM 2014, PMC3973783. Placental CYP27B1 protein vs maternal 25(OH)D.
- Ma R et al. Am J Physiol Endocrinol Metab 2012; 303:E928–35. Preeclamptic placental vitamin D metabolic component expression.
Pregnancy RCTs
- Hollis BW, Johnson D, Hulsey TC, Ebeling M, Wagner CL. J Bone Miner Res 2011; 26:2341–57. NICHD pregnancy RCT.
- Wagner CL, McNeil RB, Hamilton SA, et al. J Steroid Biochem Mol Biol 2013 (combined NICHD+Thrasher).
- Cooper C, Harvey NC, Bishop NJ, et al. (MAVIDOS Study Group). Lancet Diabetes Endocrinol 2016; 4:393–402.
- Curtis EM, Moon RJ, Harvey NC, Cooper C. JBMR 2022 — MAVIDOS 4-year follow-up.
- Litonjua AA, Carey VJ, Laranjo N, et al. JAMA 2016; 315:362–70 (VDAART).
- Wolsk HM, et al. 2023 — VDAART 15-year follow-up, J Allergy Clin Immunol.
Observational pregnancy
- Bodnar LM, Catov JM, Simhan HN, Holick MF, Powers RW, Roberts JM. J Clin Endocrinol Metab 2007. Preeclampsia.
- Bodnar LM, et al. J Nutr 2009. Bacterial vaginosis.
- Bodnar LM, Klebanoff MA, Gernand AD, et al. Epidemiology 2014; 25:207–14. Mild/severe preeclampsia.
- Achkar M, Dodds L, Giguère Y, et al. Am J Obstet Gynecol 2015; 212:511.e1–7. Preeclampsia.
- Baca KM, Simhan HN, Platt RW, Bodnar LM. J Clin Transl Endocrinol 2016; 6:75–80.
- McDonnell SL, Baggerly KA, et al. J Steroid Biochem Mol Biol 2017. 40 ng/mL & PTB.
- Tamblyn JA, Pilarski NSP, Markland AD, et al. Fertil Steril 2022. Vitamin D and miscarriage meta-analysis.
Other
- Munger KL, Levin LI, Hollis BW, Howard NS, Ascherio A. JAMA 2006; 296:2832. MS in US military.
- Munger KL, Hongell K, Åivo J, Soilu-Hänninen M, Surcel H-M, Ascherio A. Neurology 2017; 89:1578–83. MS in FMC.
- Martineau AR, Jolliffe DA, et al. BMJ 2017; 356:i6583. ARI IPD meta-analysis.
- Castillo ME, Costa LME, Vaquero Barrios JM, Alcalá Díaz JF, López Miranda J, Bouillon R, Quesada Gomez JM. J Steroid Biochem Mol Biol 2020; 203:105751. Calcifediol/COVID-19.
Recommendations
Clinical practice (decision-ready)
In pregnancy, target maternal serum 25(OH)D of 40–60 ng/mL (100–150 nmol/L) from preconception through lactation. The Hollis–Wagner program (4,000 IU/d cholecalciferol) is safe in the NICHD RCT and reaches this target in most women regardless of race. The 400 IU/d dose in standard prenatal vitamins is inadequate for placental CYP27B1 substrate provision and routinely leaves women below 30 ng/mL.
Measure 25(OH)D, never 1,25(OH)₂D, to assess vitamin D status for any non-classical indication (immune, oncologic, pregnancy, autoimmune, infection). Serum 1,25(OH)₂D is regulated by calcium homeostasis and is uninformative — and frequently misleading — for extrarenal sufficiency.
Use daily or weekly cholecalciferol, not large bolus doses for any indication where extrarenal CYP27B1 is the mechanistic target. The Martineau IPD meta-analysis (BMJ 2017) demonstrated that the protective effect of vitamin D on respiratory infections is abolished by bolus dosing — consistent with the substrate-driven model in which immune cells need a stable 25(OH)D pool, not a transient spike.
Consider calcifediol (25(OH)D₃) when rapid restoration of extrarenal substrate is needed: hospitalized acute illness, severe deficiency in pregnancy, malabsorption, obesity. Calcifediol bypasses hepatic 25-hydroxylation and achieves stable target levels within days rather than weeks.
Treat the megalin/cubilin axis as part of the picture in CKD and diabetic nephropathy — patients may need both higher 25(OH)D and consideration of activated analogs because tissue uptake is impaired even at adequate circulating levels.
Research priorities (where the field could change recommendations)
- RCT of preconception + first-trimester supplementation to a 25(OH)D ≥40 ng/mL target, powered for preeclampsia and preterm birth as co-primary outcomes.
- Head-to-head calcifediol vs cholecalciferol trials in respiratory infection, sepsis, and post-operative settings — the cleanest test of the substrate-limited extrarenal model.
- Trials stratified by baseline 25(OH)D in autoimmune disease (T1D, MS, RA) — current null trials largely enrolled vitamin-D-replete populations.
- Tissue-level confirmation in human breast/colon/prostate tumors that local 1,25(OH)₂D scales with circulating 25(OH)D in the 20–60 ng/mL range.
Thresholds that would change the recommendation
- If a well-conducted RCT of preconception supplementation to ≥40 ng/mL fails to reduce preeclampsia and preterm birth in deficient women, the placental-substrate hypothesis would require revision.
- If Mendelian-randomization studies repeatedly fail to demonstrate causality across multiple outcomes simultaneously, residual confounding becomes the more parsimonious explanation.
Caveats
Causality vs association. Most of the extrarenal evidence remains observational; multiple high-profile RCTs of cholecalciferol supplementation have been negative for primary endpoints (VITAL, D2d, MAVIDOS primary BMC, VDAART primary asthma at age 3). Strong proponents argue this is because trials enrolled vitamin-D-replete subjects and used too-low or bolus doses; skeptics argue the underlying associations are confounded by adiposity, illness, and sun exposure. The truth is probably both: the substrate-driven mechanism is real, but observational effect sizes overestimate the supplementable component.
Subgroup-and-secondary-analysis issue. Many of the most-cited results in this report (MAVIDOS winter-birth signal; McDonnell 40-ng/mL preterm-birth analysis; VDAART asthma at later follow-up; VITAL cancer-mortality secondary) are subgroup or post-hoc analyses. They are biologically plausible and internally consistent with the substrate model, but they are hypothesis-generating, not confirmatory.
Schizophrenia signal is weakening. The 2024 Finnish Maternity Cohort study (n = 1,145 cases) found no association between maternal first/early-second-trimester 25(OH)D and offspring schizophrenia. The earlier neonatal-blood-spot signal from Eyles (Denmark) may reflect a narrow critical window or population-specific deficiency rather than a general effect.
TB prevention RCTs remain negative. Despite the elegant Liu/Modlin mechanism, recent meta-analyses (6 RCTs, 15,677 subjects) show no significant reduction in TB infection or progression. The discrepancy between the cellular mechanism and clinical outcome is real and unresolved; one hypothesis is that bolus dosing predominated and that endemic populations remain below the cathelicidin-induction threshold even after supplementation.
DBP variability is a confounder. DBP haplotypes (GC1F, GC1S, GC2) differ markedly between populations (especially African vs European ancestry) and influence both total and free 25(OH)D — confounding comparisons across racial groups and possibly explaining some of the African-American TB and pregnancy-outcome disparities that low total 25(OH)D alone cannot fully explain.
Threshold heterogeneity by tissue. "40 ng/mL" is a convenient consensus inflection point but the true threshold differs by tissue and indication. Keratinocyte CYP27B1 may saturate at much lower 25(OH)D because the same cell makes its own 25(OH)D from 7-DHC; macrophage CYP27B1 in granulomatous disease can be pathologically unrestrained (sarcoidosis hypercalcemia) and serves as a counter-example showing the system can over-produce when product-inhibition is dysregulated.
Two-tailed risk. Above ~60 ng/mL the evidence base is thinner and a few signals (falls, all-cause mortality U-curves, miscarriage in one Mendelian-randomization study) suggest hormesis. The Hollis–Wagner 40–60 ng/mL pregnancy target is conservative; routinely exceeding 80 ng/mL is not supported by current evidence.
