Zinc reduces the risk of Cardiovascular Inflammation
Zinc: A metallic shield against cardiac inflammation - Feb 2026
Metallomics, mfag004, https://doi.org/10.1093/mtomcs/mfag004 UK
Zinc (Zn) is a trace element essential for the function of over 10% of the human proteome, yet the average adult body contains only about two grams. Despite its trace status, Zn plays an indispensable role in immune regulation, inflammation control, and redox signalling. Low Zn status is associated with impaired immune function and increased oxidative stress—factors that critically contribute to the pathogenesis of cardiac inflammatory diseases (CIDs), including myocarditis and pericarditis. These conditions are rising in incidence globally, particularly in younger adults, and are linked to viral infections, autoimmune triggers, and post-vaccination inflammatory responses.
Zn not only protects cysteine thiol groups from oxidation but also acts as a redox-sensitive secondary messenger via the “Redox Zinc Switch” mechanism—a key process in modulating cellular responses to oxidative stress.
In the cardiovascular system, Zn influences antioxidant defence, cytokine regulation, and membrane repair pathways, including cellular responses that are regulated by protein kinase C and metallothioneins.
Emerging evidence supports Zn supplementation as a strategy to mitigate myocardial inflammation, reduce cardiac remodelling, and improve outcomes in oxidative stress driven heart diseases. This review synthesizes current knowledge on Zn’s biochemical, immunological, and therapeutic roles in cardiac inflammation. We argue that maintaining optimal Zn levels through diet or supplementation represents a promising, accessible intervention to reduce the burden of CIDs and improve cardiovascular resilience in at-risk populations.
Zinc and cardiac inflammation: strong theory, missing trials - Claude AI Deep Research 6 minutes, 400 sources
No clinical trial has ever directly tested zinc supplementation for preventing myocarditis, pericarditis, or cardiac-specific inflammation in humans. This is the single most important finding of this review. Despite robust mechanistic evidence that zinc suppresses key inflammatory pathways implicated in cardiac inflammation—and consistent meta-analytic data showing zinc reduces systemic inflammatory biomarkers like CRP, IL-6, and TNF-α—the leap from "zinc is anti-inflammatory" to "zinc prevents myocarditis" remains entirely theoretical and unvalidated. Neither the 2025 ESC Guidelines for Myocarditis and Pericarditis nor the 2024 ACC Expert Consensus Decision Pathway mention zinc in any capacity. What does exist is a compelling but circumstantial case built from meta-analyses, animal models, and epidemiological data linking zinc deficiency to worse cardiovascular outcomes.
Meta-analyses confirm zinc lowers inflammatory markers relevant to the heart
Multiple systematic reviews and meta-analyses of randomized controlled trials consistently demonstrate that zinc supplementation reduces systemic inflammatory biomarkers central to cardiac inflammation pathophysiology. A 2021 meta-analysis of 22 adult studies found zinc reduced CRP by −0.75 mg/L (95% CI: −0.98 to −0.52; P < 0.00001). A separate meta-analysis of 25 RCTs (n = 1,428) showed IL-6 decreased by −3.81 pg/mL (P = 0.014) and malondialdehyde (a lipid peroxidation marker) decreased by −0.78 μmol/L (P < 0.001). Total antioxidant capacity increased significantly.
The most comprehensive recent review—Nazari et al. (2023), a GRADE-assessed dose-response meta-analysis of 75 RCTs—confirmed significant reductions in CRP, IL-6, TNF-α, and oxidative stress markers with zinc supplementation, alongside improvements in triglycerides, total cholesterol, and glycemic control. These findings are relevant because CRP, IL-6, and TNF-α are established mediators of myocardial inflammation. CRP is an independent predictor of cardiovascular events, IL-6 drives the "IL-6 amplifier" loop via synergistic NF-κB/STAT3 activation in cardiac tissue, and TNF-α signaling through TNFR1 promotes cardiomyocyte apoptosis, fibrosis, and contractile dysfunction.
However, the effect sizes vary substantially across populations. CRP reduction was greatest in patients with renal dysfunction (WMD: −7.43 mg/L) and at zinc doses ≥50 mg/day (WMD: −1.97 mg/L). The effect was not significant in children and was modest in healthy populations. TNF-α results are inconsistent: one meta-analysis of 21 RCTs showed significant reduction (ES = −0.49 pg/mL; P = 0.006), while another found no significant change. This heterogeneity suggests zinc's anti-inflammatory effects are dose-, duration-, and population-dependent rather than universal.
The molecular case for zinc in cardiac protection is substantial
Zinc modulates cardiac inflammation through at least five convergent molecular mechanisms, each supported by moderate-to-strong evidence from in vitro and animal studies.
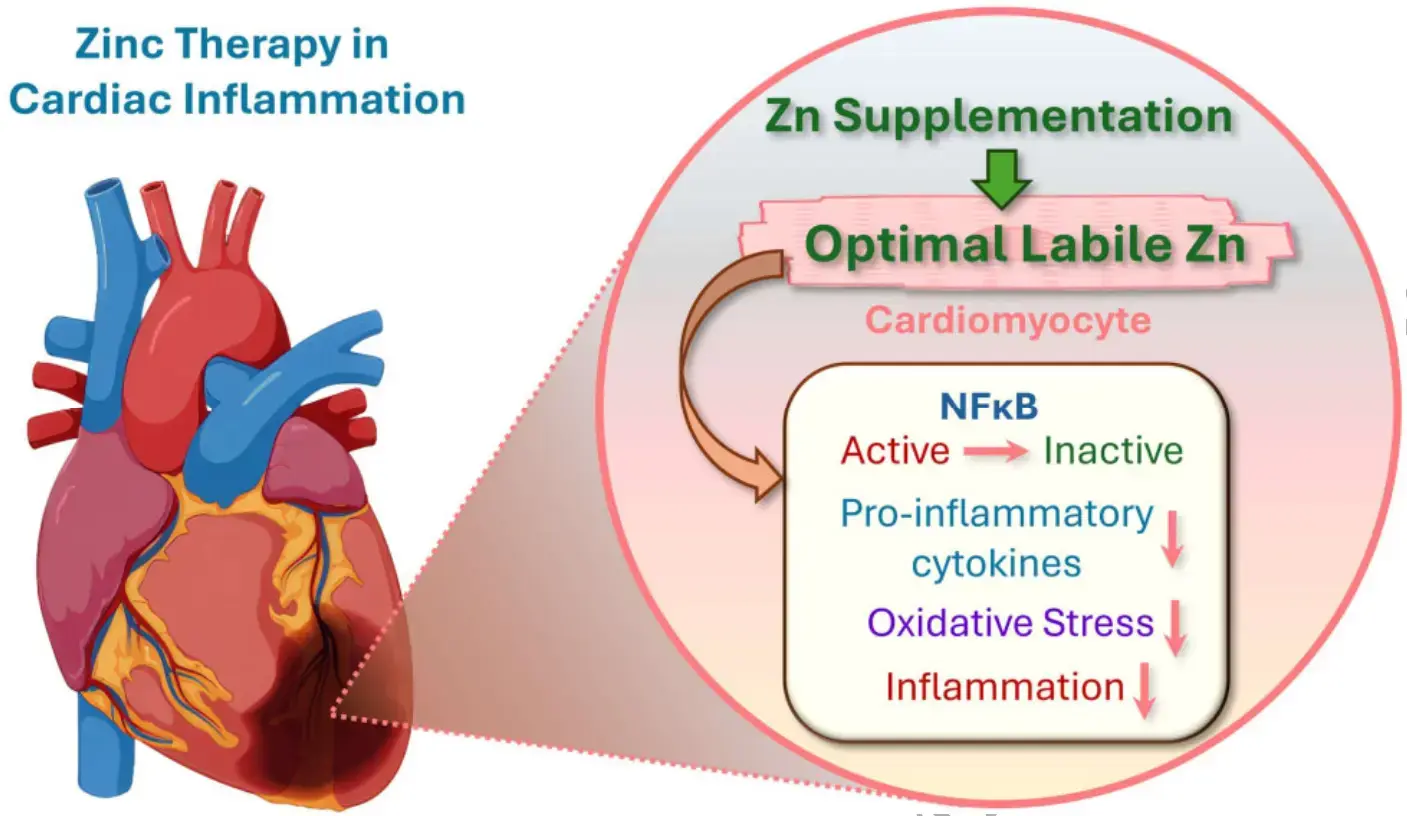
NF-κB suppression represents the best-characterized pathway. Zinc ions imported by the transporter ZIP8 directly inhibit IκB kinase (IKK), preventing NF-κB nuclear translocation. Simultaneously, zinc upregulates A20 (TNFAIP3), a zinc finger deubiquitinase that disassembles the IKK complex by removing K63-linked ubiquitin chains from RIP1, TRAF6, and NEMO. Zinc also activates PPAR-α and PPAR-γ, which further suppress inflammatory gene transcription. In human monocytic and endothelial cell lines, zinc-sufficient conditions (15 μM) dramatically reduced ox-LDL-induced TNF-α, IL-1β, VCAM-1, and NF-κB activation compared to zinc-deficient conditions (1 μM). In the heart specifically, chronic NF-κB activation is detrimental—transgenic mice with constitutive NF-κB inhibition showed reduced mortality from heart failure, fewer inflammatory cytokines, and less apoptosis.
Metallothionein induction provides a second protective mechanism. Zinc induces metallothionein (MT) synthesis via Metal Regulatory Transcription Factor 1. These cysteine-rich proteins (20 cysteine residues each) serve as direct oxidant scavengers in cardiac tissue. In cardiac-specific MT-overexpressing transgenic mice, MT overexpression improved contractile function recovery after ischemia-reperfusion, reduced infarct size, suppressed oxidative stress, and inhibited cardiomyocyte apoptosis. MT also protects against LPS-induced septic cardiac dysfunction by reducing ROS production—a finding directly relevant to inflammation-driven myocardial injury.
Antioxidant defense via Cu/Zn-superoxide dismutase (SOD1), for which zinc is a structural cofactor, and via glutathione system maintenance adds further protection. In rat myocardial ischemia-reperfusion models, acute dietary zinc deficiency exacerbated cardiac injury specifically through glutathione depletion. Chronic zinc supplementation (5 mg/kg for 15 days) markedly restored cardiac GSH levels and reduced tissue MDA. Zinc also inhibits NADPH oxidase directly—zinc pyrithione applied at reperfusion in Langendorff-perfused rat hearts improved myocardial recovery up to 100% and reduced arrhythmias more than twofold.
Immune cell modulation is particularly relevant to autoimmune and viral myocarditis. Zinc deficiency shifts macrophages toward the pro-inflammatory M1 phenotype while reducing anti-inflammatory M2 macrophages. It also disrupts Th1/Th2 balance by impairing the zinc finger transcription factor GATA-3, essential for Th2 differentiation. A 2025 study in Science Advances demonstrated that Zn²⁺ blocks macrophage pyroptosis via modulation of the lysosomal zinc pool, with anti-atherosclerotic effects. For viral myocarditis specifically, the Zinc Finger Antiviral Protein (ZAP) is induced in heart tissues of Coxsackievirus B3-infected mice, and ZAP overexpression significantly reduced cardiac inflammatory cytokine production and protected mice from acute viral myocarditis.
Antiviral activity against SARS-CoV-2 has been demonstrated in vitro. Zinc directly inhibits RNA-dependent RNA polymerase (RdRp) and the main protease (3CLpro) with a measured binding affinity (KD) of 1.965 μM. Zinc pyrithione plus 2 μM Zn²⁺ reduced SARS-CoV-GFP signal by 85% in cell culture. Since ACE2—the SARS-CoV-2 receptor expressed on cardiomyocytes—is itself a zinc metalloenzyme, zinc status could theoretically influence viral entry and downstream cardiac damage. These antiviral mechanisms are noteworthy but remain limited to in vitro systems; the NIH explicitly recommended against zinc supplementation above RDA for COVID-19 prevention due to insufficient clinical evidence.
64% of Cardiac Failure patients are zinc-deficient
Epidemiological data consistently link low zinc status to increased cardiovascular risk. The Ludwigshafen Risk and Cardiovascular Health (LURIC) Study followed 3,316 coronary angiography patients for a median 7.75 years and found that the lowest zinc quartile had 44% higher total mortality (adjusted HR: 1.44; 95% CI: 1.13–1.83) and significantly higher cardiovascular mortality (HR per quartile decrease: 1.10; P = 0.038). A 2025 multicenter retrospective study of 8,290 heart failure patients confirmed that zinc deficiency (serum Zn < 70 μg/dL) independently predicted all-cause mortality (HR: 1.46), major adverse cardiovascular events (HR: 1.46), and hospitalization (HR: 1.24) after propensity score matching.
The prevalence of zinc deficiency in cardiac populations is striking. An estimated 62–66% of heart failure patients are zinc-deficient, driven by hyperzincuria from diuretics and RAAS inhibitors, reduced dietary intake, and inflammation-mediated zinc redistribution. Among elderly individuals (the group most vulnerable to both zinc deficiency and cardiac inflammation), 30–45% have inadequate zinc status. In a retrospective study of 243 MI patients, those with serum zinc below 66 μg/dL had a 15.7-fold higher hazard for subsequent heart failure admission.
The strongest interventional evidence for zinc-deficient cardiac patients comes from Frustaci et al., who treated 10 patients with malabsorption-related dilated cardiomyopathy using intravenous zinc and selenium for six months. Left ventricular ejection fraction improved from 27% to 42% (P < 0.001), while untreated patients showed no improvement. A case report of a 24-year-old woman with anorexia nervosa and biventricular failure (serum zinc 23 μg/dL) documented LVEF improvement with zinc supplementation that reversed when zinc was discontinued and re-improved upon resumption.
The AREDS trial provides the largest long-term dataset: among ~4,757 participants randomized to 80 mg zinc oxide daily for up to 10 years, the zinc group had 27% lower all-cause mortality (RR = 0.83; 95% CI: 0.73–0.95), with the benefit driven primarily by reduced cardiovascular deaths. However, this was not the trial's primary endpoint, and confounding from antioxidant co-supplementation cannot be excluded.
A critical counterpoint exists. Seet et al. (2011) gave 240 mg/day zinc to 40 type 2 diabetic patients with normal zinc levels for three months and found no effect on oxidative damage markers or vascular function. A large Australian longitudinal study found high dietary zinc associated with higher CVD incidence in women over 50 (OR = 1.43 for highest quintile). These null and negative findings suggest zinc's cardiovascular benefits may be confined to those who are deficient, and that excess zinc could be harmful—a pattern consistent with a U-shaped dose-response curve.
Need 15-45 mg of Zinc daily if deficient
Clinical trial data converge on 15–45 mg/day elemental zinc as the effective range for anti-inflammatory and cardiovascular benefits. The landmark Prasad et al. RCTs used 45 mg/day in elderly subjects and demonstrated significant reductions in hsCRP, IL-6, MCP-1, VCAM-1, and infections (reduced by ~66%). A dose-response meta-analysis found that doses below 25 mg/day were as beneficial as higher doses for cardiovascular risk factors while avoiding potential adverse effects. The NHANES cross-sectional analysis identified a dietary zinc threshold of approximately 10.4–10.6 mg/day, beyond which additional intake provided no further CVD risk reduction.
The tolerable upper intake level set by the Institute of Medicine is 40 mg/day for adults. Doses exceeding 50 mg/day risk copper depletion (requiring 2 mg/day copper co-supplementation), and very high doses (80+ mg/day) have been associated with reduced HDL cholesterol in some studies. Zinc gluconate and zinc acetate are the most commonly used forms in clinical trials. The evidence strongly favors targeting supplementation to populations with documented or likely zinc deficiency—the elderly, heart failure patients, diabetics, and those on diuretics—rather than universal supplementation.
Interactions with vitamin D and omega-3s show theoretical synergy but limited proof
Zinc and vitamin D share a biochemical feed-forward loop: the vitamin D receptor (VDR) contains a zinc finger DNA-binding domain essential for transcriptional activation, while vitamin D regulates zinc transporters (ZIP and ZnT families) that control cellular zinc homeostasis. One RCT showed zinc supplementation for eight weeks increased vitamin D levels in postmenopausal women. A trial of combined magnesium-zinc-calcium-vitamin D supplementation in gestational diabetes patients significantly reduced hsCRP (P < 0.05) and MDA (P = 0.003) while increasing total antioxidant capacity (P = 0.01), though isolating the zinc-vitamin D interaction from other nutrients is impossible in this design.
For omega-3 fatty acids, a mechanistic synergy exists: zinc is a cofactor for delta-6-desaturase, the rate-limiting enzyme converting short-chain fatty acids to EPA and DHA. Both nutrients independently inhibit NF-κB signaling. One RCT in type 2 diabetes patients found combined omega-3 plus zinc produced significant triglyceride reduction (P < 0.001) where neither alone was significant. However, a well-designed RCT by Foster et al. (2013) testing zinc (40 mg/day) and alpha-linolenic acid in postmenopausal diabetic women found no significant effects on IL-1β, IL-6, TNF-α, or CRP after 12 weeks. A 2020 review in Biochimie argued that zinc, omega-3s, and vitamin D all share NF-κB inhibitory properties and recommended maintaining sufficiency of all three, particularly in elderly populations—but no large-scale trial has tested this triple combination for cardiac inflammation endpoints.
Conclusion
The evidence base for zinc and cardiac inflammation exists on a spectrum from strong (mechanistic and biomarker data) to absent (direct myocarditis/pericarditis prevention trials). Zinc unquestionably reduces systemic inflammatory markers in meta-analyses of RCTs, modulates NF-κB and multiple cardioprotective pathways in preclinical models, and correlates with better cardiovascular outcomes epidemiologically—especially in deficient populations. The 62–66% prevalence of zinc deficiency in heart failure patients represents a potentially correctable risk factor that current guidelines entirely overlook.
Yet the absence of any clinical trial specifically testing zinc for myocarditis or pericarditis—and the explicit absence of zinc from both the 2025 ESC and 2024 ACC myocarditis guidelines—means recommending zinc supplementation specifically for cardiac inflammation prevention would outrun the evidence. The null findings in zinc-replete populations and the potential for harm at high doses reinforce that this is not a simple "more is better" story. The most defensible position is that maintaining adequate zinc status (serum zinc ≥ 70 μg/dL) through moderate supplementation of 15–40 mg/day appears beneficial for general cardiovascular health and systemic inflammation reduction, particularly in at-risk populations, but its role in preventing specific cardiac inflammatory conditions like myocarditis and pericarditis remains a plausible hypothesis awaiting its first dedicated trial.
Related in VitaminDWiki
- Autoimmune disease treated by Vitamin D, Zinc (and other activators of Vitamin D Receptor)
- Vitamin D and Cardiovascular Health - 2025
- Heart attack risk halved in adults with heart disease if >40 ng level of vitamin D (RCT)
- Heart Failure and Vitamin D - many studies
- Cardiovascular Disease is treated by Vitamin D - many studies
- Inflammation markers reduced by Vitamin D - umbrella meta-analysis
- Inflammation
