IBD and Crohn's patients need Vitamin D, even to increase drug efficacy (Vedolizumab)
It has been known for years that Vitamin D both prevents and treats many gut problems
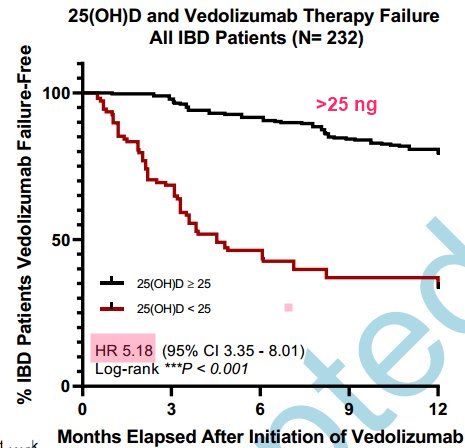
Now a study finds that a drug used to treat some gut problems (Vedolizumab) does 5X better if there is even a modest level of Vitamin D
Wonder if they have ever tried treating the gut problems by just using Vitamin D
A few charts from the study
5X better if Vitamin D > 25 ng
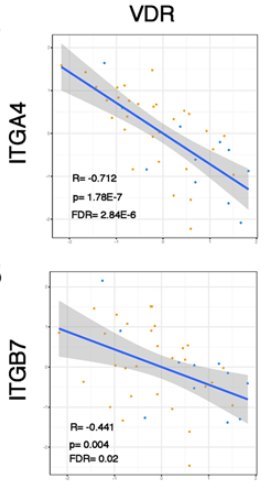
Vitamin D Receptor should also be activated
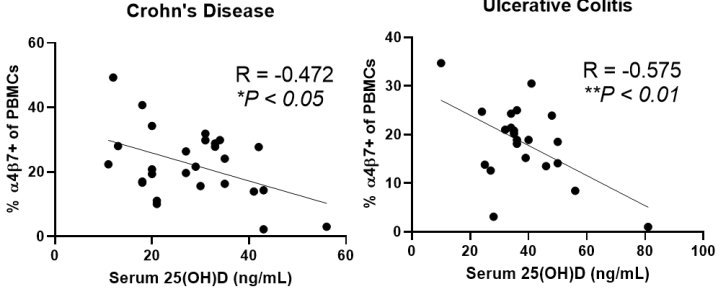
Note: Nothing special about 25 ng. The higher the vitamin D the better
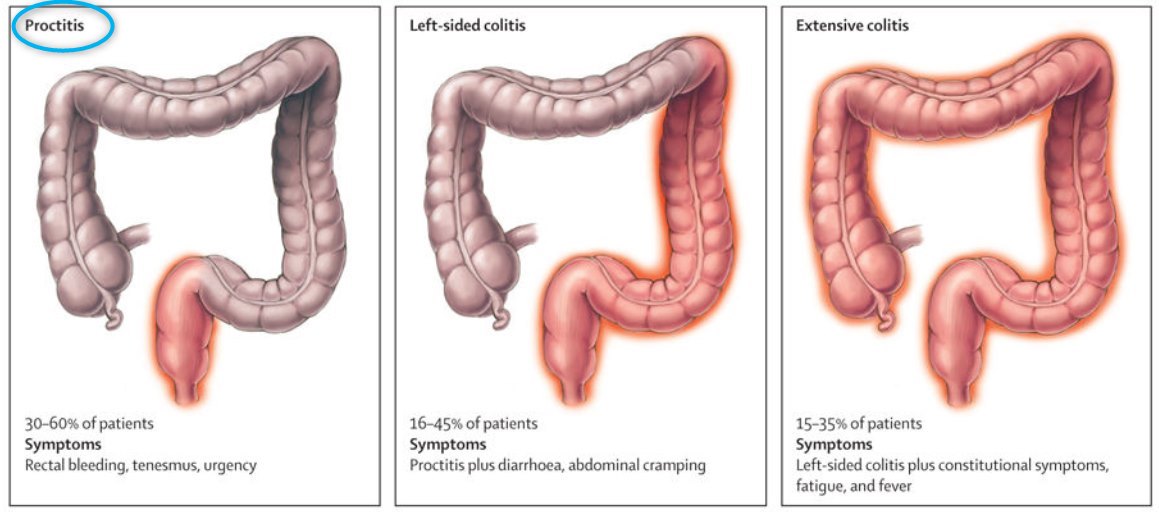 Note: Vitamin D enema works well for proctitus.
Would would need to find a time-release oral form for other UC and Crohn's
Note: Vitamin D enema works well for proctitus.
Would would need to find a time-release oral form for other UC and Crohn's
Vitamin D Levels May Predict Response to Vedolizumab - Editorial
Journal of Crohn's and Colitis, jjab105, https://doi.org/10.1093/ecco-jcc/jjab105
Giorgos Bamias, Jesús Rivera-Nieves
A remarkable expansion of therapeutic options for inflammatory bowel diseases [IBD] has arisen in recent years, mostly due to the development of monoclonal antibodies and small molecules that target inflammatory modules with presumed significance for the immunopathogenesis of disease. However, despite extraordinary progress, the long-term outcomes of novel therapies remain uniformly suboptimal, with less than half of patients achieving sustained 1-year remission on any particular treatment. This unmet need becomes even more pressing when considering the negative effect of previous treatment failures on the efficacy of subsequent medications. Precision medicine strategies have been proposed as a solution, by aiming at the sub-classification of patients into smaller, more homogeneous groups, based on the presence or absence of certain phenotype-defining biomarkers. The latter are envisioned to assist in pre-treatment selection of patients who might be most likely to respond to a particular therapy, oftentimes based on pathophysiological pathways targeted by specific drugs. In fact, increasing evidence continues to emerge reporting clinical, serological, and molecular signatures that may predict enhanced response to currently used biologics.
In this issue of JCC, Gubatan and colleagues add to that evidence by uncovering that baseline serum vitamin D [vitD] levels may serve as a predictor of therapeutic response to the anti-α4β7 integrin monoclonal antibody, vedolizumab, in patients with IBD. In particular, a value of 25[OH]D less than 25 ng/mL was associated with higher probability of primary non-response and treatment failure at 1 year. From a clinical perspective this is an important finding, given that vitD defects are highly prevalent in IBD; thus, a substantial proportion of patients would fall beneath the threshold of 25 ng/mL, rendering them prone to a suboptimal response. In the Gubatan study, 28% of patients fell in this low pre-treatment 25[OH]D group, but this percentage may be much higher in clinical practice.1 Interestingly, similar associations between baseline 25[OH]D levels and therapeutic efficacy has also been shown for anti-tumour necrosis factor [TNF] agents, whereas on-treatment increases of vitD were also noted in responders to TNF inhibitors, in both Crohn’s disease [CD] and ulcerative colitis [UC].2,3 Such evidence emphasises the need for the development of uniform definitions for vitD deficiency and insufficiency, as well as uniform recommendations for supplementation and correction of deficits.4
By definition, a biomarker needs to be simple to measure, widely available, and associated with the disease process, with ease of serial measurements. Serum vitD level fulfils those criteria, which makes it a very plausible candidate for such a role. This highly contrasts with most published work, which typically report prognostic indicators that may be cumbersome to implement. In particular, their determination requires performing repeated invasive, resource-intensive colonoscopies, evaluating on-treatment time points, analysing mucosal mRNA expression of distinct gene panels, or applying advanced technologies, all of which are largely impractical for ‘real-world’ clinical use.5–7 An added value of serum vitD level as a biomarker is that its deficits can be easily corrected. Nevertheless, the study by Gubatan does not address the important question of whether exogenous replacement of vitD may directly augment the potential response to vedolizumab. An alternative possibility is that low serum vitD content is a surrogate marker for underlying, disease-associated processes that should be corrected to maximise the potential for a therapeutic efficacy of vedolizumab.
The present study additionally provides a pathophysiological rationale in support the of the use of vitD as a biomarker for vedolizumab response, through the modification of α4β7 expression by leukocytes. In particular, the authors demonstrate an inverse correlation between serum levels of 25[OH]D and expression of α4β7 by intestinal and peripheral blood immunocytes. Furthermore, mucosal expression of vitD receptor [VDR] gene correlated negatively, whereas the VitD inactivating enzyme CYP24A1 gene correlated positively, with the expression of integrin subunits α4[ITGA4] and β7[ITGB7]. Taken together, these findings indicate that higher vitD serum concentration may decrease the expression of α4β7 by immunocytes, thus rendering them less susceptible to the inhibitory effect of vedolizumab. These data are confirmatory of previous work showing that 1,25 D3 suppressed the expression of α4β7 and CCR9 on T cells and innate lymphoid cells and antagonised the effects of the major gut-homing molecule inducer, retinoic acid.8 Nevertheless, it should be noted that when several α4β7 leukocyte subsets were separately analysed via mass cytometry, serum levels of 25[OH]D correlated with changes in integrin expression in B cells, NK cells, and monocytes, but not T cells. These current findings are in line with studies showing that responders to vedolizumab demonstrate discrete changes in innate immune cells and B lymphocytes, rather than CD4+ T cells.6,7 Thus, a paradigm shift may be taking place on our understanding of the mode of action of anti-integrin strategies, with less of an emphasis on T cell-centred biology and towards a greater emphasis of their effects on innate immune pathways and B cell biology. Given the almost universal tissue and cellular expression of VDR, the aforementioned populations are amenable to the effects of vitD and their function potentially modifiable by its deficiency. Finally, an interesting perspective may involve indirect effects of vitD via microbiota alterations, which may then affect responses to vedolizumab, as already alluded to by previous work.
Thus, should we start recommending pre-treatment evaluation of serum 25[OH]D levels in patients with IBD, based on the results presented by Gubatan et al.? If on the low side, should we consider alternatives to vedolizumab? Or, should we proactively supplement vitD with the expectation that the chance for response may increase? It may be premature to jump to such conclusions based on a single study, which should be seen as proof of concept to be validated on a larger scale and in a prospective fashion. Indeed, at the moment, a common compromising factor in biomarker research is limited reproducibility. This is oftentimes related to non-widely available state-of-the-art technologies employed in such studies. This discourages other groups to embark on cumbersome and resource-intensive validation efforts that may be then categorised by reviewers as lacking novelty. In that sense, measurement of VitD levels gives us a window of opportunity, as it is widely used in everyday clinical practice and poses no major technical challenges, and serial evaluation is feasible. An additional issue that remains is that of specificity. Are low VitD levels associated with suboptimal response specifically to vedolizumab, as supported by the evidence presented herein? Or alternatively, is it a general indicator of suboptimal response to other anti-inflammatory strategies as well, as alluded to by similar results in anti-TNF treated patients? Although the current report opens new frontiers, additional work lies ahead before the conceptual advance acquires true translational significance.
The Study
Vitamin D is Associated with α4β7+ Immunophenotypes and Predicts Vedolizumab Therapy Failure in Patients with Inflammatory Bowel Disease
J Crohns Colitis . 2021 Jun 28;jjab114. doi: 10.1093/ecco-jcc/jjab114
John Gubatan 1 2, Samuel J S Rubin 1 3, Lawrence Bai 1 3, Yeneneh Haileselassie 1, Steven Levitte 1, Tatiana Balabanis 1, Akshar Patel 1, Arpita Sharma 1, Sidhartha R Sinha 1, Aida Habtezion 1 3
Background and aims: Vitamin D downregulates the in vitro expression of the gut-tropic integrin α4β7 on immune cells. The clinical relevance of this finding in patients with inflammatory bowel disease (IBD) is unclear. We tested the hypothesis that vitamin D is associated with α4β7 immunophenotypes and risk of vedolizumab (anti- α4β7) failure in IBD.
Methods: We performed single-cell immunophenotyping of peripheral and intestinal immune cells using mass cytometry (CyTOF) in vedolizumab-naïve patients with IBD (N=48). We analyzed whole-genome mucosal gene expression (GSE73661) from GEMINI I and GEMINI long-term safety (LTS) to determine the association between vitamin D receptor (VDR) and integrin alpha-4 (ITGA4) and beta-7 (ITGB7) genes. We estimated the odds of vedolizumab failure with low pre-treatment vitamin D in a combined retrospective and prospective IBD cohort (N= 252) with logistic regression.
Results: Immunophenotyping revealed that higher 25(OH)D was associated with decreased α4β7+ peripheral blood mononuclear cells (R = -0.400, P < 0.01) and α4β7+ intestinal leukocytes (R = -0.538, P= 0.03). Serum 25(OH)D was inversely associated with α4β7+ peripheral B cells and natural killer (NK) cells and α4β7+ intestinal B cells, NK cells, monocytes, and macrophages. Mucosal expression of VDR was inversely associated with ITGA4 and ITGB7 expression. In multivariate analysis, 25(OH)D < 25 ng/mL was associated with increased vedolizumab primary non-response during induction (OR 26.10, 95% CI 14.30-48.90, P<0.001) and failure at 1-year follow-up (OR 6.10, 95% CI 3.06-12.17, P<0.001).
Conclusions: Low serum 25(OH)D is associated with α4β7+ immunophenotypes and predicts future vedolizumab failure in patients with IBD.
