Summary of Vitamin D trials - Grant
Original Jan 2012 - updated Nov 2012
CLICK HERE for web page which has Jan 2012 table and associated references & abstracts
Nov 2012 document with full references is attached at the bottom of this page
List of random controlled vitamin D trials covered in version of Jan 2012:
{SPLIT(colsize=220|220|320)}
All cause mortality
Atopic dermatitis
Bone loss in winter
Breast cancer
Colorectal cancer
Cancer, All
Cardiometric
Cardiovascular
Cardiovascular disease event
Chronic kidney disease
CVD survival
Depression
Falls
Falls, fractures
Gastric bypass
Glucose (fasting plasma) (FPG)
Haemodialysis
Heart failure, congestive
Infections, symptomatic upper respiratory tract
Infectious diseases
Influenza
Influenza type A
Insulin sensitivity
Multiple sclerosis
Muscle strength
Musculoskeletal parameters
Neuromuscular
Non-vertebrate fractures
Physical performance
Pneumonia
Pregnancy
Stress fractures
Testosterone
Tooth loss
Tuberculosis
{SPLIT}
See also by Dr. Grant
Vitamin D overview by Grant - Jan 2011 - Requirements for Vitamin D Across the Life Span
Strength of vitamin D evidence – Dr. Grant letter to editor July 2012
Evidence that the minimum vitamin D blood level is 30 ng – Grant Nov 2012
|
Outcome |
Conditions |
Finding |
Reference |
|
All
cause mortality |
Mean oral in take 528 IU/d |
RR = 0.93
(95% CI, 0.87-0.99) |
Autier and Gandini, 2007 |
|
|
daily vitamin D(3) (800
IU), calcium (1000 mg), both, or placebo for 24-62 months, with a follow-up
of 3 yr after intervention |
(HR) = 0.93; 95% confidence interval (CI) =
0.85-1.02] |
Avenell, 2012 |
|
All
cause mortality |
|
Vitamin D with or without calcium reduced mortality by 7%
[hazard ratio, 0.93; 95% confidence interval (CI), 0.88-0.99]. However, vitamin D alone did not affect mortality, but risk of
death was reduced if vitamin D was given with
calcium (hazard ratio, 0.91; 95% CI, 0.84-0.98). The number needed to treat
with vitamin D plus calcium for 3 yr to prevent
one death was 151. Trial level meta-analysis (24 trials with 88,097
participants) showed similar results, i.e. mortality was reduced with vitamin D plus calcium (odds ratio, 0.94; 95% CI,
0.88-0.99), but not with vitamin D alone (odds
ratio, 0.98; 95% CI, 0.91-1.06). |
Rejnmark, 2012 |
|
Atopic
dermatitis |
|
|
Sidbury, 2008 |
|
Bone
loss in winter |
oral cholecalciferol (500 IU/day) and calcium (500
mg/day) during the winter months |
In the subjects receiving oral vitamin D3 and
calcium, lumbar and femoral BMD increased significantly (lumbar spine: +0.8%,
p = 0.04 versus year 1; femoral neck: +0.1%, p = 0.05 versus year 1), whereas
controls continued to lose bone (intervention group versus control group:
lumbar spine, p = 0.03; femoral neck, p = 0.05). |
Meier, 2004 |
|
Cancer,
All |
1100 IU/d, 1450 mg/d calcium |
When analysis was confined to cancers diagnosed
after the first 12 mo, RR for the Ca + D group fell to 0.232 (CI: 0.09, 0.60;
P < 0.005) but did not change significantly for the Ca-only group. |
Lappe, 2007 |
|
|
daily vitamin D(3) (800
IU), calcium (1000 mg), both, or placebo for 24-62 months, with a follow-up
of 3 yr after intervention |
cancer mortality (HR = 0.85; 95% CI = 0.68-1.06),
and cancer incidence (HR = 1.07; 95% CI = 0.92-1.25) |
Avenell, 2012 |
|
Breast,
colorectal cancer |
400 IU/d vitamin D3, 1500 mg/d or placebo |
In 15,646 women (43%) who were not taking personal
calcium or vitamin D supplements at
randomization, CaD significantly decreased the risk of total, breast, and
invasive breast cancers by 14-20% and nonsignificantly reduced the risk of
colorectal cancer by 17%. In women taking personal calcium or vitamin D supplements, CaD did not alter cancer risk
(HR: 1.06-1.26). |
Bolland, 2011 |
|
Cardiometric |
In meta-analyses of 10 trials |
supplementation nonsignificantly reduced systolic
blood pressure (weighted mean difference, -1.9 mm Hg [CI, -4.2 to 0.4 mm Hg])
and did not affect diastolic blood pressure (weighted mean difference, -0.1
mm Hg [CI, -0.7 to 0.5 mm Hg]). |
Pittas, 2010 |
|
Cardiovascular |
Vitamin D supplementation during weight loss |
more pronounced decrease occurred in the vitamin D
group than in the placebo group in blood concentrations of parathyroid
hormone (-26.5% compared with -18.7%; P = 0.014), triglycerides (-13.5%
compared with +3.0%; P < 0.001), and the inflammation marker tumor
necrosis factor-alpha (-10.2% compared with -3.2%; P = 0.049). |
Zittermann,
2009 |
|
Cardiovascular
disease event |
Pooled study |
.Results of secondary analyses in 8 randomized
trials showed a slight but statistically nonsignificant reduction in CVD risk
(pooled relative risk, 0.90 [95% CI, 0.77 to 1.05]) with vitamin D supplementation at moderate to high doses
(approximately 1000 IU/d) but not with calcium supplementation (pooled
relative risk, 1.14 [CI, 0.92 to 1.41]), or a combination of vitamin D and calcium supplementation (pooled relative
risk, 1.04 [CI, 0.92 to 1.18]) compared with placebo. |
Wang, 2010 |
|
CVD |
Participants
were randomized 2:1 to vitamin D3 4,000 IU daily or placebo for 12 weeks. The
primary outcome was a change in flow-mediated brachial artery dilation (FMD). |
Baseline demographics were similar except for age (vitamin D versus placebo, mean ±sd 47 ±8 versus 40 ±10
years; P=0.009). Both groups had reduced FMD at baseline (median values 2.9%
[IQR 1.6-4.8] for vitamin D versus 2.5% [IQR
1.7-6.4] for placebo; P=0.819). Despite an increase in the concentration of
serum 25-hydroxyvitamin D from baseline to 12 weeks (5.0 ng/ml [IQR -0.9-7.4]
versus -1.9 ng/ml [IQR -4.0-0.1] for vitamin D
versus placebo, respectively; P=0.003), there was no difference in FMD change
(0.55% [IQR -1.05-2.13] versus 0.29% [IQR -1.61-1.77]; P=0.748). Vitamin D supplementation was associated with a
decrease in total and non-high-density lipoprotein cholesterol, and an
increase in indices of insulin resistance. |
Longenecker, 2012 |
|
CVD
survival |
|
Vitamin D supplementation
conferred substantial survival benefit (odds ratio for death 0.39, 95%
confidence interval 0.28 to 0.53, p <0.0001). |
Vacek, 2012 |
|
Chronic
kidney disease |
Placebo
or calcium (1,200 mg) and vitamin D(3) (800 IU) in fixed or separate
combination. Effect on parathyroid hormone |
The proportion with a 30% or greater decrease in
iPTH level at 6 months was 50% in all eGFR groups on treatment versus 6% to
9% for placebo (P < 0.001 for all). The effects of the intervention on
iPTH levels did not differ according to baseline eGFR (interaction P > 0.1
for all times). |
Kooienga, 2009 |
|
|
1000 IU/d vs. placebo |
Mean 25(OH)D levels increased significantly higher
in the treatment group (mean increase from baseline: 10.3+/-10.4 ng/mL vs.
0.8+/-6.8 ng/mL, p<0.0001). |
Rucker, 2009 |
|
|
Successful 25OHD replacement was defined as
prescription of ergocalciferol sufficient to increase serum 25OHD level by
25% from baseline within 6 months (treatment group, n = 90). Otherwise
patients were considered as untreated controls (n = 36). The date when the
25OHD level was drawn was considered as the date of inclusion. All the
patients were followed up from the date of inclusion until July 2009 to
capture CV events prospectively. |
During mean follow-up of 27.2 months, 44% of the
controls had CV events, whereas only 21% of the patients in the treatment
group had CV events (P = 0.001). In multivariate logistic regression
analysis, adjusting for CV disease predictors
age, initial parathyroid hormone level, statin use, history of CV disease, and glomerular filtration rate, the estimated
odds ratio for 25OHD replacement status was 0.37 (95% confidence interval:
0.14-1.0). |
Lishmanov, 2012 |
|
Depression |
Cross-sectional study and randomized double blind
controlled trial of 20,000 or 40,000 IU vitamin D per week versus placebo for
1 year. |
In the two groups given vitamin D, but not in the
placebo group, there was a significant improvement in BDI scores after 1
year. There was a significant decrease in serum parathyroid hormone in the
two vitamin D groups without a concomitant increase in serum calcium. |
Jorde, 2008 |
|
Falls,
fractures |
700-1000 IU/d |
Pooled RR = 0.81 (95% CI 0.71 to 0.92) No effect for 400 IU/d |
Bischoff-Ferrari, 2009b |
|
Falls |
Our study population consisted of 242 individuals
recruited by advertisements and mailing lists (mean [ +/- SD] age, 77 +/- 4
years). All serum 25-hydroxyvitamin D (25[OH]D) levels were below 78 nmol/l.
Individuals received in a double blinded fashion either 1000 mg of calcium or
1000 mg of calcium plus 800 IU of vitamin D per day over a treatment period
of 12 months, which was followed by a treatment-free but still blinded
observation period of 8 months. |
Compared to calcium mono, supplementation with
calcium plus vitamin D resulted in a significant decrease in the number of subjects
with first falls of 27% at month 12 (RR = 0.73; CI = 0.54-0.96) and 39% at
month 20 (RR = 0.61; CI = 0.34-0.76). Concerning secondary endpoints, we observed
significant improvements in quadriceps strength of 8%, a decrease in body
sway of 28%, and a decrease in time needed to perform the TUG test of 11%. |
Pfeifer, 2009 |
|
Non-vertebrate
fractures |
700-1000 IU/d |
pooled RR was 0.80 (95% CI, 0.72-0.89; |
Bischoff-Ferrari, 2009a |
|
Fractures |
Participants who were randomly assigned to receive
vitamin D, as compared with those assigned to control groups, |
had a nonsignificant 10% reduction in the risk of
hip fracture (hazard ratio, 0.90; 95% confidence interval [CI], 0.80 to 1.01)
and a 7% reduction in the risk of nonvertebral fracture (hazard ratio, 0.93;
95% CI, 0.87 to 0.99). By quartiles of actual intake, reduction in the risk
of fracture was shown only at the highest intake level (median, 800 IU daily;
range, 792 to 2000), with a 30% reduction in the risk of hip fracture (hazard
ratio, 0.70; 95% CI, 0.58 to 0.86) and a 14% reduction in the risk of any
nonvertebral fracture (hazard ratio, 0.86; 95% CI, 0.76 to 0.96). |
Bischoff-Ferrari, 2012 |
|
Flow-mediated
dilation |
Following 16 weeks of placebo (n = 23; mean age 31 ±
2 years) or 60,000 IU monthly oral vitamin D(3)
(n = 22; mean age 29 ± 2 years), serum concentrations of 25-hydroxyvitamin D
(25(OH)D) increased from 38.2 ± 3.0 to 48.7 ± 3.2 nmol/l and 34.3 ± 2.2 to
100.9 ± 6.6 nmol/l, respectively. |
No changes in serum parathyroid hormone (PTH), serum calcium, or
urine calcium/creatinine were observed following
either treatment. Following 16 weeks of treatment, significant improvements
in FMD were only observed in the vitamin D group
(1.8 ± 1.3%), whereas the placebo group had no change (-1.3 ± 0.6%).
Similarly, the vitamin D group exhibited an
increase in absolute change in diameter (0.005 ± 0.004 cm) and FMD/shear
(0.08 ± 0.04 %/s(-1), area under the curve (AUC) × 10(3)) following
treatment, whereas no change (-0.005 ± 0.002 cm and -0.02 ± 0.02 %/s(-1),
AUC, respectively) was observed following placebo. |
Harris, 2011 |
|
Gastric
bypass |
We evaluated three doses of
vitamin D3 (800, 2,000, and 5,000 IU/day) in a prospective, randomized pilot
trial of 45 patients undergoing Roux-en-Y gastric bypass. |
At 12 months, the 800-,
2,000-, and 5,000-IU groups had a mean +/- SD increase in 25OHD of 27.5 +/-
40.0, 60.2 +/- 37.4, and 66.1 +/- 42.2 nmol/L, respectively (p = 0.09) with a
maximum increase in each group of 87.4, 114.8, and 129.8 nmol/L. Forty-four
percent, 78%, and 70% achieved 25OHD levels >or=75 nmol/L (p = 0.38). |
Goldner, 2009 |
|
Glucose
(fasting plasma) (FPG) |
A total of 314 Caucasian adults without diabetes
received either 500 mg calcium citrate and 700 IU vitamin
D(3) or placebos daily for 3 years in a double-blind, randomized,
controlled trial designed for bone-related outcomes. |
Among participants with IFG at baseline, those who
took combined calcium-vitamin D supplements had
a lower rise in FPG at 3 years compared with those on placebo (0.02 mmol/l
[0.4 mg/dl] vs. 0.34 mmol/l [6.1 mg/dl], respectively, P = 0.042) and a lower
increase in HOMA-IR (0.05 vs. 0.91, P = 0.031). |
Pitas, 2007 |
|
Haemodialysis |
HD patients were included in this study if their
serum 25(OH)D level was <75 mmol/L. Oral 25(OH)D(3) was administered daily
at 10-30 microg/day based on the severity of the deficiency. |
After 6 months of treatment [mean daily 25(OH)D(3):
16 +/- 5 microg/day], the serum 25(OH)D level increased (30 +/- 19 to 126 +/-
46 nmol/ L, P < 0.001), with 13% of patients in group 1, 57% in group 2
and 30% in group 3. The serum intact parathyroid hormone (iPTH) level
decreased (235 +/- 186 to 189 +/- 137 pg/mL, P = 0.05), except in group 1.
Bone alkaline phosphatase (BALP) showed a tendency to normalize (23 +/- 16 to
18.3 +/- 11 microg/L, P < 0.05), leading to a decrease in alfacalcidol
administration from 66% to 43% (P < 0.05), except in group 1. The KDOQI
targets achieved increased significantly for serum calcium (76% to 85%) and
phosphate levels (66% to 77%) in all patients. The serum albumin level
increased in all groups (34.6 +/- 4 to 36.8 +/- 4 g/L, P < 0.05), without
any significant improvement in normalized protein catabolic rate (nPCR) or
C-reactive proteins (CRP). |
Jean, 2008 |
|
Heart
failure, congestive |
either 50 mug vitamin D(3)/d
plus 500 mg Ca/d [D(+) group] or placebo plus 500 mg Ca/d [D(-) group] for 9
mo |
Compared
with baseline, parathyroid hormone was significantly lower and the
antiinflammatory cytokine interleukin 10 was significantly higher in the D(+)
group after 9 mo. The proinflammatory cytokine tumor necrosis factor alpha
increased in the D(-) group but remained constant in the D(+) group. The
survival rate did not differ significantly between the study groups during
the follow-up period. |
Schleithoff, 2006 |
|
Infectious
diseases |
|
|
Yamshchikov, 2009 |
|
Infections,
symptomatic upper respiratory tract |
50 microg vitamin D3 (2000 IU) daily or matching
placebo for 12 weeks. |
The mean 25-hydroxyvitamin D level at baseline was
similar in both groups (64.3+/-25.4 nmol/l in the vitamin D group; 63.0+/-25.8
nmol/l in the placebo group; n.s.). After 12 weeks, 25-hydroxyvitamin D
levels increased significantly to 88.5+/-23.2 nmol/l in the vitamin D group,
whereas there was no change in vitamin D levels in the placebo group. There
was no benefit of vitamin D3 supplementation in decreasing the incidence or
severity of symptomatic URIs during winter. |
Li-Ng, 2009 |
|
Inflammation |
|
Vitamin D
status was the sole determinant of circulating MMP9 (inversely) and an
independent determinant of CRP (inversely). Determinants of TIMP-1 were MMP9,
systolic blood-pressure (directly) and VDR genotype (TaqI). Significant
reductions in MMP9 (-68%), TIMP-1 (-38%) and CRP (-23%) concentrations
followed vitamin-D supplementation. |
Timms, 2002 |
|
|
2000 IU/d vitamin D3 given to diabetics |
Vitamin
D deficiency was common, affecting 36% of patients. There was no impact of
hypovitaminosis D on metabolic syndrome status, HbA1c or insulin use (p
>or= 0.4 for all) and no association between 25OHVitD and ferritin or
hsCRP (p >or= 0.3 for all). Neither BMI nor the metabolic syndrome
affected the incremental rise in 25OHVitD levels during supplementation. |
Luo, 2009 |
|
Influenza |
800 or 2000 IU/d |
40 or 90% reduction |
Aloia & Li-Ng, 2007 |
|
Influenza
type A |
1100 IU/d, no other oral intake |
RR: 0.36; 95% CI: 0.17, 0.79; P = 0.006 No effect for type B |
Urashima, 2010 |
|
Insulin
sensitivity |
120,000 IU/fortnight |
Seventy-one of the recruited subjects completed the
study (35 in supplemented group, 36 in control group). There was an increase
in oral glucose insulin sensitivity (OGIS) with supplementation by per
protocol analysis (P = 0.038; intention-to-treat analysis P = 0.055). The
age- and baseline 25-hydroxyvitamin D level-adjusted difference in change in
OGIS was highly significant (mean difference 41.1 +/- 15.5; P = 0.01). No
changes in secondary outcome measures (insulin secretion, basal indices of
insulin sensitivity, blood pressure or lipid profile) were found with
supplementation. |
Nagpal, 2009 |
|
|
double-blind intervention administering 100 microg
(4000 IU) vitamin D(3) (n 42) or placebo (n 39) daily for 6 months to South
Asian women, aged 23-68 years, living in Auckland, New Zealand |
Significant improvements were seen in insulin
sensitivity and IR (P = 0.003 and 0.02, respectively), and fasting insulin
decreased (P = 0.02) with supplementation compared with placebo. |
Von hurst, 2010 |
|
|
Only
subjects in the intervention group received one intramuscular injection of
300,000 IU of vitamin D3. HbA(1c), serum 25-hydroxyvitamin D3, fasting
insulin and blood glucose, C-peptide, homeostasis model assessment insulin
resistance index (HOMA-IR), β-cell function, insulin sensitivity and the
Quantitative Insulin Sensitivity Check Index (QUICKI) were measured at
baseline and after 3 months of intervention. |
Approximately
80% of the mothers had a degree of vitamin D deficiency. Post-intervention,
this was found in 4.2 and 71.4% in the intervention and control groups,
respectively. The medians of HOMA-IR indices before and after intervention
were 0.6 and 0.5 (P = 0.7), respectively, in subjects in the intervention
group, and 0.5 and 0.9 (P = 0.01) in subjects in the control group. The mean
of the QUICKI fell only in the control group (P = 0.008). In the control
group, β-cell function increased by ~8% (P = 0.01) and insulin
sensitivity decreased after 3 months (P = 0.002). Post-intervention, the
median C-peptide decreased in the intervention group and increased in the
control group, but the change was significant only in the control group (P =
0.03). |
Mozaffari-Khosravi, 2012 |
|
|
participants with low serum 25(OH)D levels were
thereafter randomized to receive capsules of 20,000 IU vitamin
D(3) or identical-looking placebo twice weekly for 6 months. |
The 52 participants with high serum 25(OH)D levels
(85.6 ± 13.5 nmol/L [mean ± SD]) had significantly higher insulin sensitivity
index (ISI) and lower HbA(1c) and triglycerides (TGs) than the 108
participants with low serum 25(OH)D (40.3 ± 12.8 nmol/L), but the differences
in ISI and TGs were not significant after adjustments. After supplementation,
serum 25(OH)D was 142.7 ± 25.7 and 42.9 ± 17.3 nmol/L in 49 of 51 completing
participants randomized to vitamin D and 45 of
53 randomized to placebo, respectively. At the end of the study, there were
no statistically significant differences in the outcome variables between the
two groups. |
Grimnes, 2011 |
|
|
cholecalciferol (2000 IU once daily) or calcium
carbonate (400 mg twice daily) for 16 wk. |
The disposition index increased in the vitamin D group and decreased in the no-vitamin D group (adjusted mean change ± SE: 300 ± 130
compared with -126 ± 127, respectively; P = 0.011), which was explained by an
improvement in insulin secretion (62 ± 39 compared with -36 ± 37 mU · L(-1) ·
min, respectively; P = 0.046). Hb A(1c) increased less, but nonsignificantly,
in the vitamin D group than in the no-vitamin D group (0.06 ± 0.03% compared with 0.14 ±
0.03%, respectively; P = 0.081). |
Mitri, 2011 |
|
Muscle
strength |
6-month supplementation
(December to May) of daily calcium plus monthly placebo (calcium/placebo
group) or daily calcium plus oral cholecalciferol (150,000 IU once a month
during the first 2 months, followed by 90,000 IU once a month for the last 4
months; calcium/vitamin D group) |
SHF
was increased in the calcium/vitamin D group by 16.4% (p = 0.0001) and SKE by
24.6% (p = 0.0007). [strength of hip flexors
(SHF) and knee extensors (SKE)] |
Moreira-Pfrimer, 2009 |
|
|
800 or 1600 IU/d plus vibration |
Same improvements with both doses |
Verschueren, 2011 |
|
|
Twenty-six (87%) patients completed 3 months of
therapy. The percent increase in serum 25OHD (compared to baseline) was 82.7%
in 2,000-IU group and 219.5% in 5,000-IU group. All participants (100%)
achieved a serum 25OHD concentration >50 nmol/L; only 5 subjects
(45.4%) in 2,000-IU group compared to 14 subjects (93.3%) in 5,000-IU group
achieved final 25OHD concentration ≥75 nmol/L (p < 0.01). |
In the regression analysis, the reflexion point at
which the PTH level increased above the normal range was calculated to be
63.8 nmol/L 25OHD. All parameters of muscle strength showed trends in
improvements following the administration of both the 2,000 and 5,000 IU
doses. No patient reported untoward side effects and no patient developed |
Diamond, 2012 |
|
Musculoskeletal
parameters |
One hundred seventy-nine girls, ages 10-17 yr, were
randomly assigned to receive weekly oral vitamin D
doses of 1,400 IU (equivalent to 200 IU/d) or 14,000 IU (equivalent to 2,000
IU/d) in a double-blind, placebo-controlled, 1-yr protocol. |
In the overall group of girls, lean mass increased
significantly in both treatment groups (P < or = 0.05); bone area and
total hip BMC increased in the high-dose group (P < 0.02). In
premenarcheal girls, lean mass increased significantly in both treatment
groups, and there were consistent trends for increments in BMD and/or BMC at
several skeletal sites, reaching significance at lumbar spine BMD in the
low-dose group and at the trochanter BMC in both treatment groups. There was
no significant change in lean mass, BMD, or BMC in postmenarcheal girls. |
El-Hajj, 2006 |
|
Multiple
sclerosis |
6000 IU/d vitamin D2 vs. 1000
IU/d vitamin D2 |
There were 4 relapses with high-dose D2 vs none with
low-dose D2 (p = 0.04). |
Stein, 2011 |
|
Neuromuscular |
8400 IU/week |
significantly reduced sway compared with treatment
with placebo (P = 0.047) in patients with elevated baseline sway but not in
patients with normal baseline sway. |
Lips, 2010 |
|
Periodontal
disease |
A cohort of 51 subjects receiving periodontal maintenance therapy was recruited from two
dental clinics; 23 were taking vitamin D
(>or=400 IU/day) and calcium (>or=1,000 mg/day) supplementation, and 28
were not taking such supplementation. All subjects had at least two
interproximal sites with >or=3 mm clinical attachment loss. Daily calcium
and vitamin D intake (from food and supplements)
were estimated by nutritional analysis. |
Compared to subjects who did not take vitamin D and calcium supplementation, supplement
takers had shallower probing depths, fewer bleeding sites, lower gingival
index values, fewer furcation involvements, less attachment loss, and less
alveolar crest height loss. The repeated-measures analysis indicated that
collectively these differences were borderline significant (P = 0.08). |
Miley, 2009 |
|
Physical
performance |
|
gait speed was higher among subjects supplemented
with vitamin (whether trained or not) than in non-supplemented subjects
(838+/-147 and 768+/-127 m/12 min, respectively, p=0.02). |
Bunout, 2006 |
|
Pneumonia |
Children, 100,000 IU D3 once |
Children
in the vitamin D(3) group survived longer
without experiencing a repeat episode (72 days vs. 59 days; HR 0.71; 95% CI
0.53-0.95; P = 0.02). |
Manaseki-Holland, 2010 |
|
|
Oral vitamin D (1000 IU
for <1 year and 2000 IU for >1 year) (n=100) or placebo (lactose)
(n=100) once a day for 5 days, from enrolment. |
Median duration (SE, 95% CI) of resolution of severe
pneumonia was similar in the two groups [vitamin D:
72 (3.7, 64.7-79.3) hours; placebo: 64 (4.5, 55.2-72.8)hours]. Duration of
hospitalization and time to resolution of tachypnea, chest retractions, and
inability to feed were also comparable between the two groups. |
Choudhary & Gupta, 2011 |
|
Pregnancy |
4000 IU/d |
No adverse effects on serum or urine calcium |
Hollis, 2011 |
|
|
Maternal 25(OH)D (n = 161) increased from 22.7 ng/mL
(SD 9.7) at baseline to 36.2 ng/mL (SD 15) and 37.9 ng/mL (SD 13.5) in the
2000 and 4000 IU groups, respectively. While maternal 25(OH)D change from
baseline did not differ between groups, 25(OH)D monthly increase differed
between groups (P < .01). No supplementation-related adverse events
occurred. Mean cord blood 25(OH)D was 22.1 ± 10.3 ng/mL in 2000 IU and 27.0 ±
13.3 ng/mL in 4000 IU groups (P = .024). |
After
controlling for race and study site, preterm birth and labor were inversely
associated with predelivery and mean 25(OH)D, but not baseline 25(OH)D. |
Wagner, 2012 |
|
Respiratory
infections |
At baseline, the median serum 25(OH)D level was 7
ng/mL (interquartile range: 5-10 ng/mL). At the end of the trial, follow-up
was 99% (n = 244), and the median 25(OH)D levels of children in the control
versus vitamin D groups was significantly
different (7 vs 19 ng/mL; P < .001). |
Compared
with controls, children receiving vitamin D
reported significantly fewer ARIs during the study period (mean: 0.80 vs
0.45; P = .047), with a rate ratio of 0.52 (95% confidence interval:
0.31-0.89). Adjusting for age, gender, and history of wheezing, vitamin D continued to halve the risk of ARI (rate
ratio: 0.50 [95% confidence interval: 0.28-0.88]). Similar results were found
among children either below or above the median 25(OH)D level at baseline
(rate ratio: 0.41 vs 0.57; P(interaction) = .27). |
Camargo, 2012 |
|
Stress
fractures |
Navy females, 800 IU/d, 2000 mg/d calcium |
21% lower incidence |
Lappe, 2008 |
|
Testosterone |
Participants received either 83 μg (3,332 IU)
vitamin D daily for 1 year (n = 31) or placebo (n =2 3). |
Compared to baseline
values, a significant increase in total testosterone levels (from 10.7 ± 3.9
nmol/l to 13.4 ± 4.7 nmol/l; p < 0.001), bioactive testosterone (from 5.21
± 1.87 nmol/l to 6.25 ± 2.01 nmol/l; p = 0.001), and free testosterone levels
(from 0.222 ± 0.080 nmol/l to 0.267 ± 0.087 nmol/l; p = 0.001) were observed
in the vitamin D supplemented group. By contrast, there was no significant
change in any testosterone measure in the placebo group |
Pilz, 2011 |
|
Tooth
loss |
Elderly, taking supplements or not |
During the randomized trial, 11 of the 82 subjects
(13%) taking supplements and 17 of the 63 subjects (27%) taking placebo lost
one or more teeth (OR = 0.4; 95% CI: 0.2 to 0.9). |
Krall, 2001 |
|
Tuberculosis |
The subjects were randomised to receive vitamin D
(0.25 mg/day) or placebo in a double blind method, during the 6th initial
week of Tb treatment. |
One
hundred percent of the vitamin D group and only 76.7% of the placebo group
had sputum conversion. This difference is statistically significant
(p=0.002). |
Nursyam, 2006 |
|
|
The intervention was 100,000 IU of cholecalciferol
or placebo at inclusion and again 5 and 8 months after the start of
treatment. |
Overall mortality was 15% (54
of 365) at 1 year of follow-up and similar in both arms (30 of 187 for vitamin D treated and 24 of 178 for placebo; relative
risk, 1.19 [0.58-1.95]). |
Wejse, 2009 |
|
|
A single oral dose of 2.5 mg (100,000 IU) vitamin D |
significantly enhanced the ability of participants'
whole blood to restrict BCG-lux luminescence in vitro without affecting
antigen-stimulated IFN-gamma responses. |
Martineau, 2011 |
|
|
At
baseline, the mean (±SD) 25(OH)D concentration was 7 ± 4 ng/mL, and all
concentrations were <20 ng/mL. Vitamin D supplementation increased serum
25(OH)D by a mean of 12.7 ng/mL compared with placebo (P < 0.0001). At
baseline, 16 children in the vitamin D group and 18 in the placebo group were
TST positive (P = 0.7). |
Over
6 mo, TSTs converted to positive in 5 (11%) children receiving vitamin D
compared with 11 (27%) receiving placebo (RR: 0.41; 95% CI: 0.16, 1.09; P =
0.06). Only one TST conversion occurred among those whose serum 25(OH)D
concentration increased to >20 ng/mL, whereas 8 TST conversions occurred in
those whose final 25(OH)D concentration remained <10 ng/mL (P = 0.05). The
mean increase in stature was 2.9 ± 1.6 cm in the vitamin D group and 2.0 ±
1.7 cm in the placebo group (95% CI: 2.16, 2.81; P < 0.003). |
Ganmaa, 2012 |
See also web
- WikiPedia on Clinical Trials nothing about vitamin D
See also VitaminDWiki
Over 1000 Clinical Trials with Vitamin D INTERVENTION- Aug 2013
101 vitamin D INVERVENTION trials added during first 7 months of 2012
Proof that Vitamin D Works 38 items as of Dec 2013
Search VitaminDWiki for "W * grant" OR "William grant" 433 items as of Jan 2014
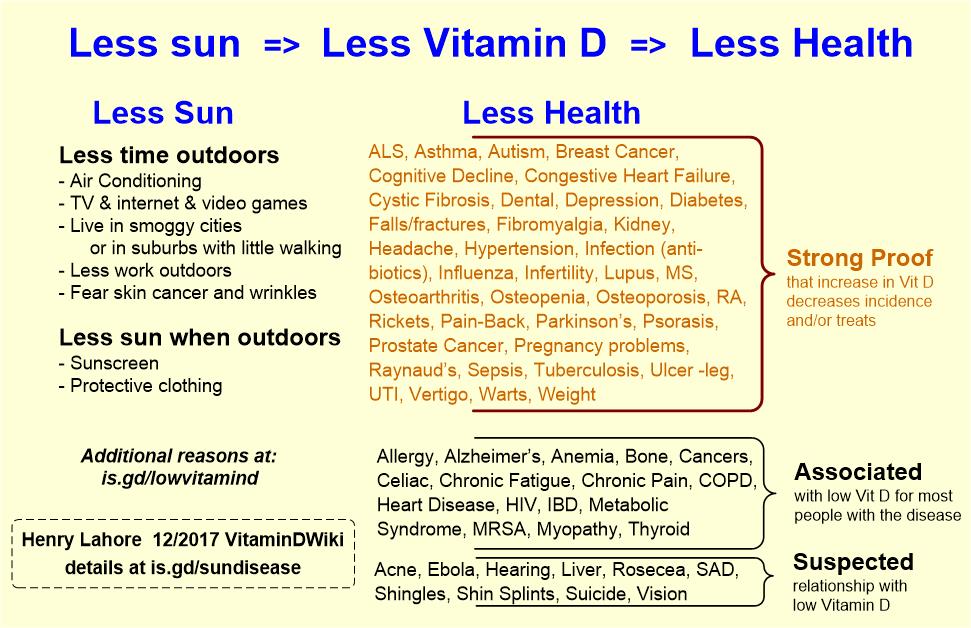
Hypothesis of cause and effect of vitamin D deficiency which has the following chart